Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The total number of carbon atoms present in tyrosine, an amino acid, is _____
Correct Answer: 9
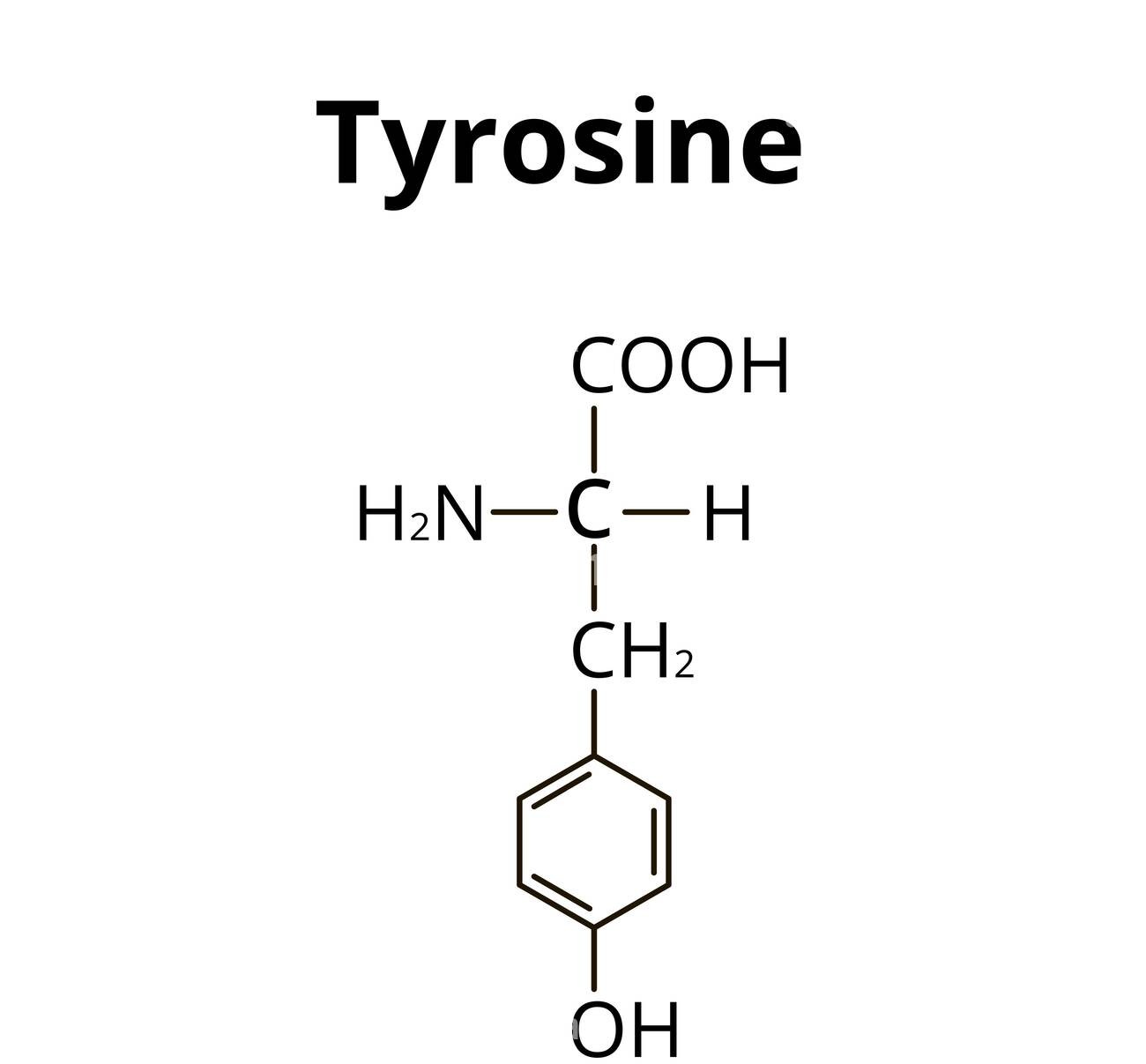
We need to find the total number of carbon atoms in tyrosine.
Tyrosine is an amino acid with the molecular formula C9H11NO3.
Its structure is 4-hydroxyphenyl alanine: it has a benzene ring (6 carbons) with an OH group, connected via a -CH2- group to the amino acid backbone -CH(NH2)-COOH.
Counting carbon atoms:
- Benzene ring: 6 carbons
- CH2- group: 1 carbon
- alpha-carbon (CH): 1 carbon
- Carboxyl group (COOH): 1 carbon
Total = 6 + 1 + 1 + 1 = 9 carbon atoms.
The answer is 9

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
