Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Match the LIST-I with LIST-II
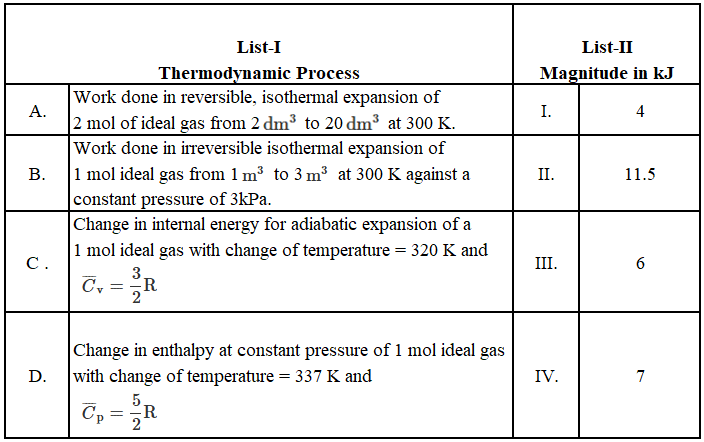
Choose the correct answer from the options given below:
For a reversible isothermal process:
$$|W| = nRT \ln\left(\frac{V_2}{V_1}\right)$$
For expansion against a constant external pressure:
$$|W| = P_{ext}(V_2 - V_1)$$
The change in internal energy ($$\Delta U$$) is calculated as:
$$\Delta U = n C_v \Delta T$$
The change in enthalpy ($$\Delta H$$) is calculated as:
$$\Delta H = n C_p \Delta T$$
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
