Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
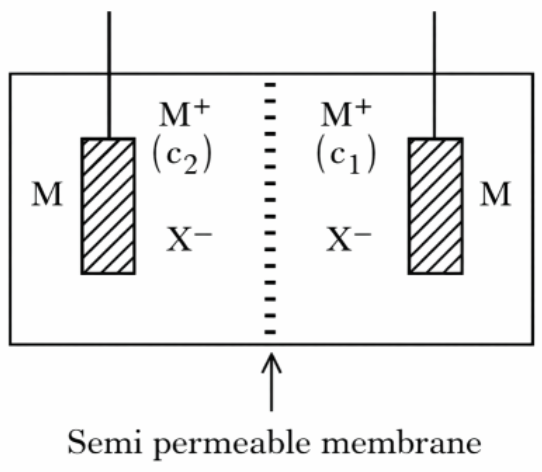
Consider the above electrochemical cell where a metal electrode (M) is undergoing redox reaction by forming $$M^{+}(M\rightarrow M^{+}+e^{-})$$. The cation $$M^{+}$$ is present in two different concentrations $$c_{1}$$ and $$c_{2}$$ as shown above. which of the following statement is correct for generating a positive cell potential?
Create a FREE account and get:
Educational materials for JEE preparation
