Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
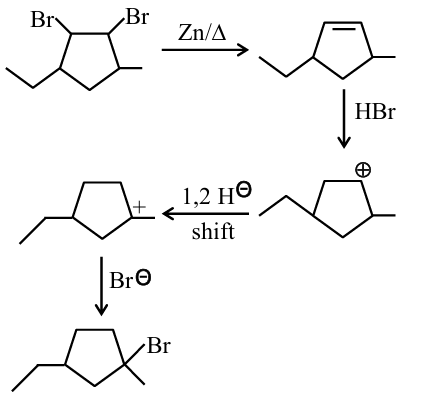
Step 1: Debromination with ($$Zn$$, $$\Delta$$)The reaction of a vicinal dibromide (1,2-dibromo compound) with zinc dust and heat undergoes an elimination reaction. The zinc removes the two bromine atoms to form a carbon-carbon double bond.
Step 2: Hydrobromination with ($$HBr$$)The intermediate alkene then reacts with ($$HBr$$). This is an electrophilic addition reaction following Markovnikov's rule.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
