Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
For strong electrolyte $$\lambda_m$$ increases slowly with dilution and can be represented by the equation
$$\Lambda_m = \Lambda_m^\circ - A c^{1/2}$$
Molar conductivity values of the solutions of strong electrolyte AB at 18°C are given below:
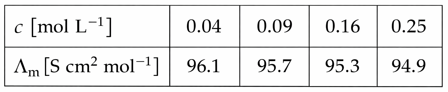
The value of constant A based on the above data [in S $$cm^{2}mol^{-1}/(mol/L)^{1/2}$$]unit is_______.
Correct Answer: 4
Create a FREE account and get:
Educational materials for JEE preparation
