Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The pH and conductance of a weak acid (HX) was found to be 5 and $$4\times10^{-5}S$$. respectively. The conductance was measured under standard condition using a cell where the electrode plates having a surface area of 1 $$cm^{2}$$ were at a distance of 15 cm apart. The value of the limiting molar conductivity is ______ S $$m^{2}mol^{-1}$$ (nearest integer)
(Given : degree of dissociation of the weak acid ($$\alpha$$) < < 1)
Correct Answer: 6
Given: Weak acid HX has pH = 5 and conductance $$G = 4 \times 10^{-5}$$ S (standard conditions).
Electrode area $$A = 1$$ cm$$^2 = 10^{-4}$$ m$$^2$$, electrode distance $$l = 15$$ cm $$= 0.15$$ m.
Cell constant = $$\frac{l}{A} = \frac{0.15}{10^{-4}} = 1500$$ m$$^{-1}$$.
Specific conductivity: $$\kappa = G \times \frac{l}{A} = 4 \times 10^{-5} \times 1500 = 0.06$$ S m$$^{-1}$$.
Since pH = 5, $$[H^+] = 10^{-5}$$ M. For weak acid HX with $$\alpha \ll 1$$: $$[H^+] = c\alpha \approx 10^{-5}$$ M.
Molar conductivity at this concentration: $$\Lambda_m = \frac{\kappa}{c}$$ where $$c$$ is in mol/m$$^3$$.
Since $$\alpha \ll 1$$, the total concentration $$c \gg [H^+]$$. However, we can use:
$$\Lambda_m = \alpha \cdot \Lambda_m^0$$ (limiting molar conductivity)
So $$\Lambda_m^0 = \frac{\Lambda_m}{\alpha} = \frac{\kappa}{c\alpha}$$.
Since $$c\alpha = [H^+] = 10^{-5}$$ mol/L $$= 10^{-2}$$ mol/m$$^3$$:
$$\Lambda_m^0 = \frac{\kappa}{c\alpha \text{ (in mol/m}^3\text{)}} = \frac{0.06}{10^{-2}} = 6$$ S m$$^2$$ mol$$^{-1}$$.
The answer is 6.
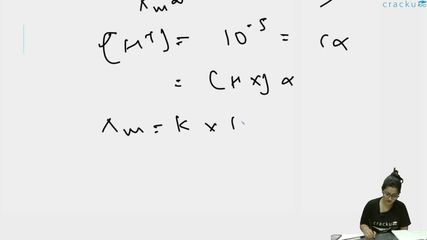
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
