Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
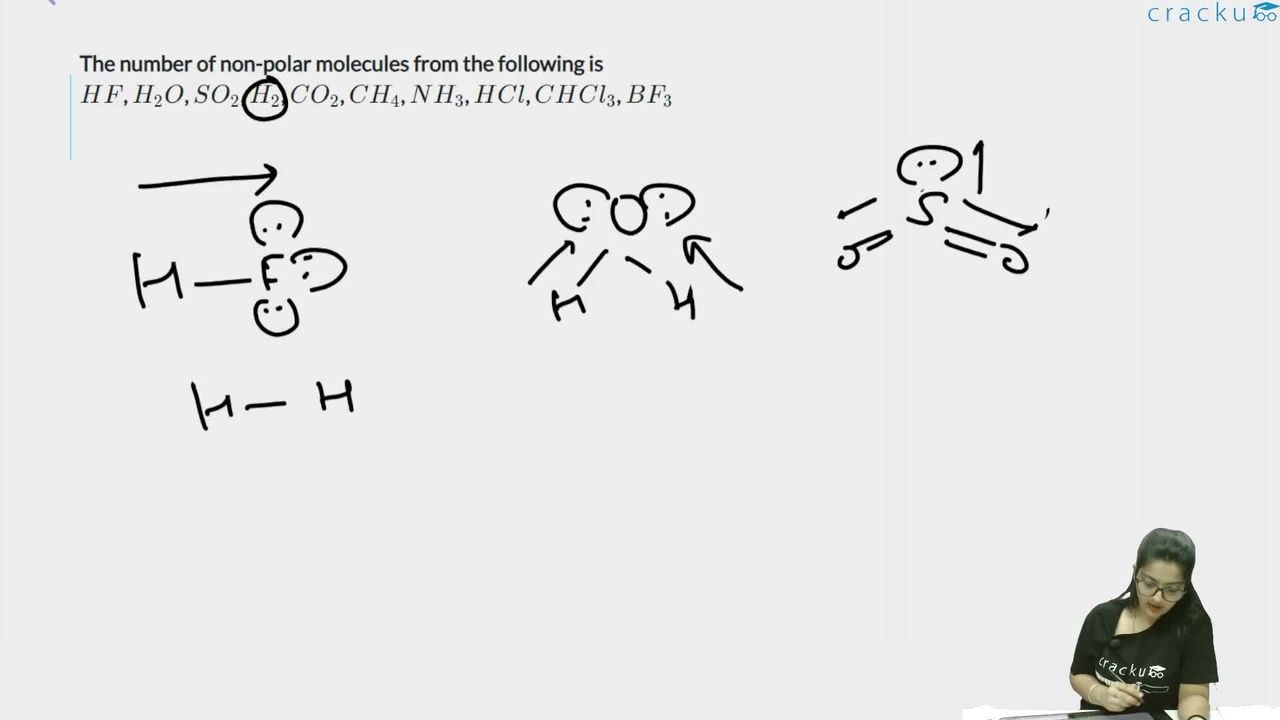
The number of non-polar molecules from the following is
$$HF$$, $$H_2O$$, $$SO_2$$, $$H_2$$, $$CO_2$$, $$CH_4$$, $$NH_3$$, $$HCl$$, $$CHCl_3$$, $$BF_3$$
Correct Answer: 4
We need to count the non-polar molecules from the given list: HF, H$$_2$$O, SO$$_2$$, H$$_2$$, CO$$_2$$, CH$$_4$$, NH$$_3$$, HCl, CHCl$$_3$$, BF$$_3$$.
A molecule is non-polar if either (a) it has no polar bonds, or (b) it has polar bonds but they are arranged symmetrically so that the dipole moments cancel out.
Analyze each molecule:
(i) HF: Polar bond, asymmetric → Polar
(ii) H$$_2$$O: Polar O-H bonds, bent geometry → dipoles don't cancel → Polar
(iii) SO$$_2$$: Polar S=O bonds, bent geometry → Polar
(iv) H$$_2$$: Same atoms, no dipole → Non-polar ✓
(v) CO$$_2$$: Polar C=O bonds, but linear and symmetric → dipoles cancel → Non-polar ✓
(vi) CH$$_4$$: Polar C-H bonds, but tetrahedral symmetry → dipoles cancel → Non-polar ✓
(vii) NH$$_3$$: Polar N-H bonds, pyramidal geometry → Polar
(viii) HCl: Polar bond, asymmetric → Polar
(ix) CHCl$$_3$$: Asymmetric substitution → dipoles don't cancel → Polar
(x) BF$$_3$$: Polar B-F bonds, but trigonal planar symmetry → dipoles cancel → Non-polar ✓
Count non-polar molecules: H$$_2$$, CO$$_2$$, CH$$_4$$, BF$$_3$$ = 4.
The answer is 4.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
