Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Sum of bond order of CO and $$NO^+$$ is _______.
Correct Answer: 6
We need to find the sum of bond orders of CO and $$NO^+$$.
We first determine the bond order of CO using Molecular Orbital Theory (MOT). The formula for bond order is:
$$ \text{Bond Order} = \frac{N_b - N_a}{2} $$
where $$N_b$$ = number of electrons in bonding molecular orbitals, and $$N_a$$ = number of electrons in antibonding molecular orbitals.
CO has a total of $$6 + 8 = 14$$ electrons. The molecular orbital configuration for CO (following the order for molecules with atomic number $$\leq 7$$ for the lighter atom) is:
$$ \begin{aligned} &(\sigma 1s)^2 (\sigma^* 1s)^2 (\sigma 2s)^2 (\sigma^* 2s)^2\\ &(\pi 2p_x)^2 (\pi 2p_y)^2 (\sigma 2p_z)^2 \end{aligned} $$
Counting electrons gives bonding electrons $$N_b = 2 + 2 + 2 + 2 + 2 = 10$$ (from $$\sigma 1s, \sigma 2s, \pi 2p_x, \pi 2p_y, \sigma 2p_z$$) and antibonding electrons $$N_a = 2 + 2 = 4$$ (from $$\sigma^* 1s, \sigma^* 2s$$). Thus,
$$ \text{Bond Order of CO} = \frac{10 - 4}{2} = \frac{6}{2} = 3 $$
Next, we determine the bond order of $$NO^+$$. This species has $$7 + 8 - 1 = 14$$ electrons (nitrogen has 7, oxygen has 8, minus 1 for the positive charge). Since $$NO^+$$ is isoelectronic with CO, it has the same MO configuration:
$$ \begin{aligned} &(\sigma 1s)^2 (\sigma^* 1s)^2 (\sigma 2s)^2 (\sigma^* 2s)^2\\ &(\pi 2p_x)^2 (\pi 2p_y)^2 (\sigma 2p_z)^2 \end{aligned} $$
Therefore,
$$ \text{Bond Order of } NO^+ = \frac{10 - 4}{2} = \frac{6}{2} = 3 $$
Finally, the sum of bond orders is
$$ \text{Sum of bond orders} = 3 + 3 = 6 $$
The answer is $$\boxed{6}$$.
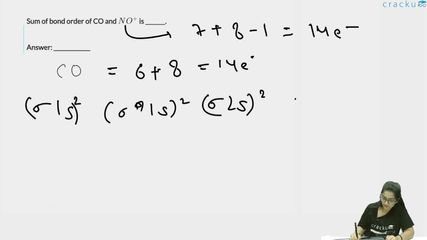
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
