NTA JEE Main 10th April 2019 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 31
The minimum amount of O$$_2$$(g) consumed per gram of reactant is for the reaction:
(Given atomic mass: Fe = 56, O = 16, Mg = 24, P = 31, C = 12, H = 1)
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 32
The ratio of the shortest wavelength of two spectral series of hydrogen spectrum is found to be about 9. The spectral series are:
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 33
The correct order of the first ionization enthalpies is:
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 34
Points I, II and III in the following plot respectively correspond to (V$$_{mp}$$: most probable velocity)
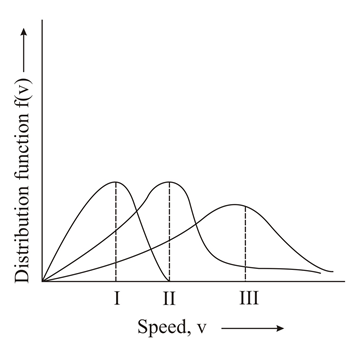
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 35
The difference between ΔH and ΔU is ΔH - ΔU, when the combustion of one mole of heptane l is carried out at a temperature T, is equal to:
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 36
For the reaction,
2SO$$_2$$g + O$$_2$$g $$\rightleftharpoons$$ 2SO $$_3$$g,
ΔH = -57.2 kJ mol$$^{-1}$$ and K$$_c$$ = $$1.7 \times 10^{16}$$.
Which of the following statements is incorrect?
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 37
The pH of a 0.02 M NH$$_4$$Cl solution will be [Given: K$$_b$$ NH$$_4$$OH = 10$$^{-5}$$ and log 2 = 0.301]
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 38
The correct statements among a to d are:
a. Saline hydrides produce H$$_2$$ gas when reacted with H$$_2$$O.
b. Reaction of LiAlH$$_4$$ with BF$$_3$$ leads to B$$_2$$H$$_6$$.
c. PH$$_3$$ and CH$$_4$$ are electron-rich and electron-precise hydrides, respectively.
d. HF and CH$$_4$$ are called as molecular hydrides.
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 39
A hydrated solid X on heating initially gives a monohydrated compound Y. Y upon heating above 373 K leads to an anhydrous white powder Z. X and Z, respectively, are;
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 40
The number of pentagons in C$$_{60}$$ and trigons (triangles) in white phosphorus, are;
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 41
The increasing order of nucleophilicity of the following nucleophiles is:
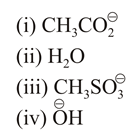
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 42
In chromatography, which of the following statements is incorrect for R$$_f$$?
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 43
Which of these factors does not govern the stability of a conformation in acyclic compounds?
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 44
The major product obtained in the given reaction is:
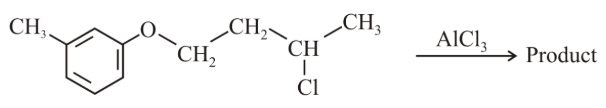
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 45
Air pollution that occurs in sunlight is
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 46
The noble gas that does not occur in the atmosphere is;
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 47
1 g of a non-volatile non-electrolyte solute is dissolved in 100 g of two different solvents A and B whose ebullioscopic constants are in the ratio of 1:5. The ratio of the elevation in their boiling points, $$\frac{\Delta T_{bA}}{\Delta T_{bB}}$$, is:
(assuming they have the same molar mass)
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 48
Which one of the following graphs between molar conductivity $$\Lambda_m$$ versus $$\sqrt{C}$$ is correct?
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 49
For the reaction of H$$_2$$ with I$$_2$$, the rate constant is $$2.5 \times 10^{-4}$$ dm$$^3$$ mol$$^{-1}$$ s$$^{-1}$$ at 327°C and 1.0 dm$$^3$$ mol$$^{-1}$$ s$$^{-1}$$ at 527°C. The activation energy for the reaction, in kJ mol$$^{-1}$$ is:
R = 8.314 JK$$^{-1}$$ mol$$^{-1}$$
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 50
The correct option among the following is.
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 51
The correct statement is:
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 52
The highest possible oxidation states of uranium and plutonium, respectively, are
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 53
The incorrect statement is.
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 54
The crystal field stabilization energy (CFSE) of [FeH$$_2$$O$$_6$$]Cl$$_2$$ and K$$_2$$[NiCl$$_4$$], respectively, are:
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 55
The major product 'Y' in the following reaction is:
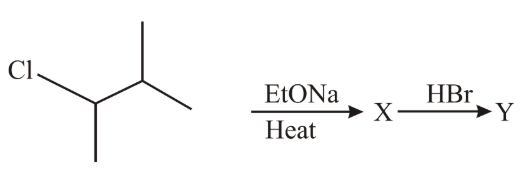
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 56
Compound A C$$_9$$H$$_{10}$$O shows positive iodoform test. Oxidation of A with KMnO$$_4$$/KOH gives acid B C$$_8$$H$$_6$$O$$_4$$. Anhydride of B is used for the preparation of phenolphthalein. Compound A is:
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 57
The major product Y in the following reaction is:
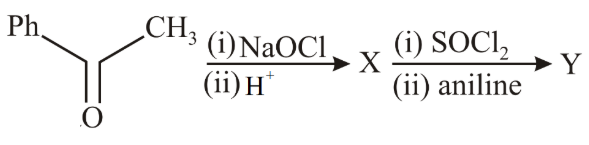
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 58
Which of the following is not a correct method of the preparation of benzyl amine from cyano benzene?
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 59
The correct match between Item - I and Item - II is:
Item - I Item - II
(a) High density polythene (I) Peroxide catalyst
(b) Polyacrylonitrile (II) Condensation at high temperature & pressure
(c) Novolac (III) Ziegler-Natta catalyst
(d) Nylon 6 (IV) Acid or base catalyst
NTA JEE Main 10th April 2019 Shift 2 - Chemistry - Question 60
Number of stereo centers present in linear and cyclic structures of glucose are respectively:
.webp)
.webp)
.webp)
.webp)




