Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
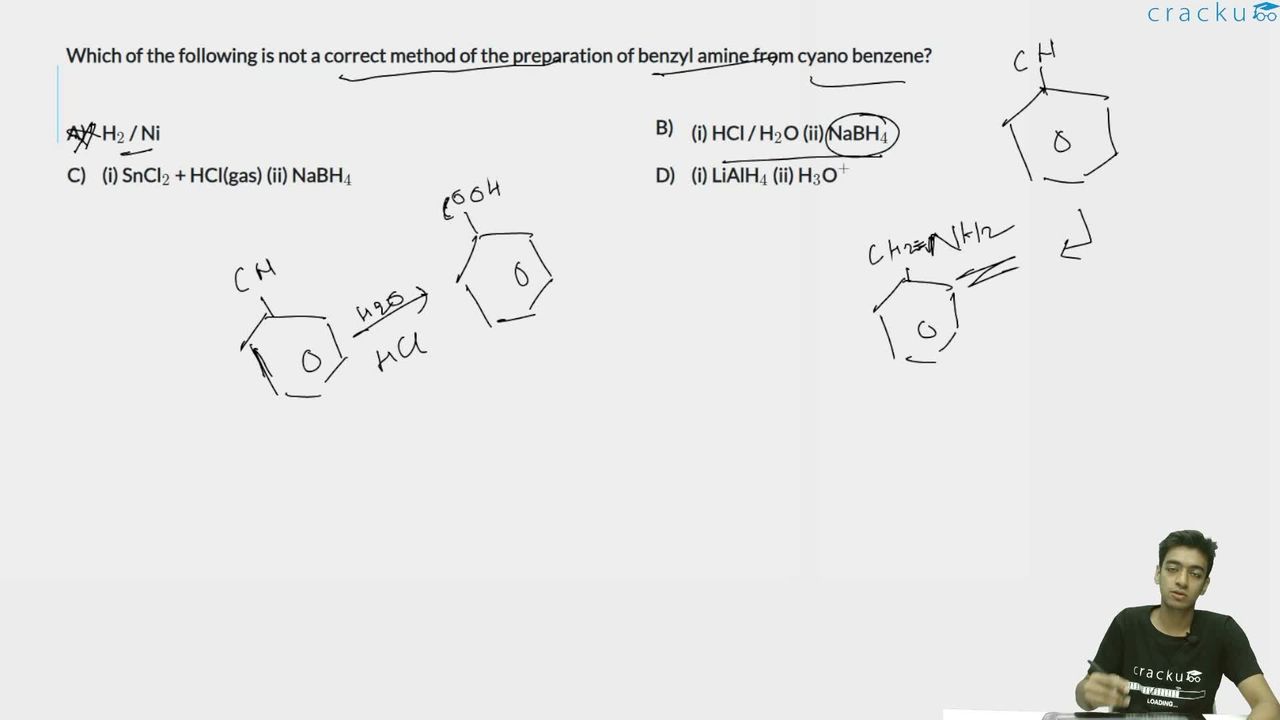
Which of the following is not a correct method of the preparation of benzyl amine from cyano benzene?
We start with cyano benzene, whose structure can be written as $$\mathrm{C_6H_5-C\equiv N}$$. The task is to see which set of reagents fails to convert this - CN group into the - CH$$_2$$NH$$_2$$ group of benzylamine $$\bigl(\mathrm{C_6H_5-CH_2NH_2}\bigr)$$.
Option A : $$\mathrm{H_2/Ni}$$
Hydrogen in the presence of a nickel catalyst carries out catalytic hydrogenation. For a nitrile, the general reaction is
$$\mathrm{R{-}C\equiv N \;+\;2\,H_2 \xrightarrow{\;Ni\;} R{-}CH_2NH_2}$$
Putting $$\mathrm{R=C_6H_5}$$ we obtain $$\mathrm{C_6H_5-CH_2NH_2}$$ directly. So Option A gives benzylamine correctly.
Option B : (i) $$\mathrm{HCl/H_2O}$$ (ii) $$\mathrm{NaBH_4}$$
Step (i) is simply acid-water hydrolysis. The - CN group is hydrolysed all the way to the carboxylic acid:
$$\mathrm{C_6H_5-C\equiv N \;+\;2\,H_2O \xrightarrow{HCl} C_6H_5-COOH \;+\;NH_4Cl}$$
Now we have benzoic acid $$\bigl(\mathrm{C_6H_5-COOH}\bigr)$$. Step (ii) employs $$\mathrm{NaBH_4}$$. At ordinary temperatures $$\mathrm{NaBH_4}$$ is able to reduce only aldehydes and ketones; it does not reduce carboxylic acids. Therefore
$$\mathrm{C_6H_5-COOH \xrightarrow{NaBH_4} \;No\;Reaction}$$
Since the - COOH group is not reduced, an amine cannot be produced. Thus Option B cannot give benzylamine.
Option C : (i) $$\mathrm{SnCl_2 + HCl(g)}$$ (ii) $$\mathrm{NaBH_4}$$
With nitriles, the Stephen reduction sequence is operative. In the first step, stannous chloride in dry hydrochloric acid converts the nitrile into an iminium chloride salt:
$$\mathrm{C_6H_5-C\equiv N \;+\,2\,HCl \;+\;SnCl_2 \;\longrightarrow\; C_6H_5-CH{=}NH\cdot HCl \;+\;SnCl_4}$$
This iminium chloride resembles an aldehyde in reactivity. In the second step $$\mathrm{NaBH_4}$$ delivers hydride, reducing the C=N bond to C-NH$$_2$$:
$$\mathrm{C_6H_5-CH{=}NH\cdot HCl \;+\; NaBH_4 \;\longrightarrow\; C_6H_5-CH_2NH_2 \;+\;NaCl \;+\;BH_3}$$
So Option C successfully furnishes benzylamine.
Option D : (i) $$\mathrm{LiAlH_4}$$ (ii) $$\mathrm{H_3O^+}$$
Lithium aluminium hydride is a strong hydride donor that reduces nitriles all the way to the amine anion. The general equation is
$$\mathrm{R{-}C\equiv N \;+\;4\,[H] \xrightarrow{LiAlH_4} R{-}CH_2NH^{-}AlH_3}$$
and after acidic work-up
$$\mathrm{R{-}CH_2NH^{-}AlH_3 \;+\;H_3O^+ \;\longrightarrow\; R{-}CH_2NH_2 \;+\;Al(OH)_3}$$
Hence Option D correctly gives benzylamine.
Summarising, Options A, C and D convert cyano benzene to benzylamine, whereas Option B stops at the acid stage and therefore fails.
Hence, the correct answer is Option B.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
