NTA JEE Main 10th April 2019 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 31
At 300 K and 1 atmospheric pressure, 10 mL of a hydrocarbon required 55 mL of O$$_2$$ for complete combustion, and 40 mL of CO$$_2$$ is formed. The formula of the hydrocarbon is:
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 32
The graph between $$|\psi|^2$$ and r (radical distance) is shown below. This represents:
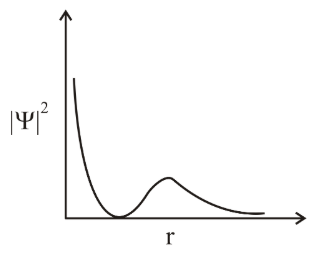
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 33
The isoelectronic set of ions is:
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 34
During the change of O$$_2$$ to O$$_2^-$$, the incoming electron goes to the orbital:
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 35
Consider the following table:
Gas a/(kPa dm$$^6$$ mol$$^{-1}$$) b/(dm$$^3$$ mol$$^{-1}$$)
A 642.32 0.05196
B 155.21 0.04136
C 431.91 0.05196
D 155.21 0.4382
a and b are van der Waals constants. The correct statement about the gases is:
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 36
A process will be spontaneous at all temperatures if:
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 37
Consider the following statements
(a) The pH of a mixture containing 400 mL of 0.1 M H$$_2$$SO$$_4$$ and 400 mL of 0.1 M NaOH will be approximately 1.3.
(b) Ionic product of water is temperature dependent.
(c) A monobasic acid with K$$_a$$ = 10$$^{-5}$$ has a pH = 5. The degree of dissociation of this acid is 50%.
(d) The Le Chatelier's principle is not applicable to common-ion effect.
The correct statements are:
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 38
The synonym for water gas when used in the production of methanol is:
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 39
The alloy used in the construction of aircrafts is:
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 40
The correct order of catenation is:
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 41
The principle of column chromatography is:
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 42
The increasing order of the reactivity of the following compounds towards electrophilic aromatic substitution reactions is:
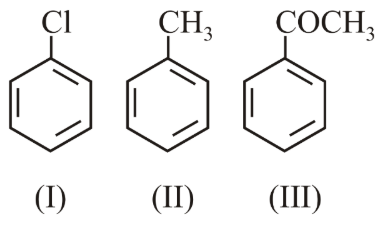
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 43
The regions of the atmosphere, where clouds form and where we live, respectively, are:
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 44
At room temperature, a dilute solution of urea is prepared by dissolving 0.60 g of urea in 360 g of water. If the vapour pressure of pure water at this temperature is 35 mm Hg, lowering of vapour pressure will be:
(molar mass of urea = 60 g mol$$^{-1}$$)
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 45
Consider the statements S1 and S2:
S1: Conductivity always increases with decreases in the concentration of electrolyte.
S2: Molar conductivity always increases with decreases in the concentration of electrolyte.
The correct option among the following
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 46
A bacterial infection in an internal wound grows as N'(t) = N$$_0$$exp(t), where the time t is in hours. A dose of antibiotic, taken orally, needs 1 hour to reach the wound. Once it reaches there, the bacterial population goes down as $$\frac{dN}{dt} = -5N^2$$. What will be the plot of $$\frac{N_0}{N}$$ vs t after 1 hour?
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 47
A gas undergoes physical adsorption on a surface and follows the given Freundlich adsorption isotherm equation
$$\frac{x}{m} = kp^{0.5}$$
Adsorption of the gas increases with:
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 48
Match the refining methods (Column I) with metals (Column II).
Column I Column II
(Refining methods) (Metals)
(I) Liquation (a) Zr
(II) Zone Refining (b) Ni
(III) Mond Process (c) Sn
(IV) Van Arkel Method (d) Ga
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 49
The oxoacid of sulphur that does not contain bond between sulphur atom is:
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 50
Consider the hydrated ions of Ti$$^{2+}$$, V$$^{2+}$$, Ti$$^{3+}$$ and Sc$$^{3+}$$. The correct order of their spin-only magnetic moments is:
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 51
The species that can have a trans-isomer is:
(en = ethane-1, 2-diamine, ox = oxalate)
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 52
Three complexes,
[CoCl(NH$$_3$$)$$_5$$]$$^{2+}$$(I), [Co(NH$$_3$$)$$_5$$H$$_2$$O]$$^{3+}$$(II) and [Co(NH$$_3$$)$$_6$$]$$^{3+}$$(III)
absorb light in the visible region. The correct order of the wavelength of light absorbed by them is:
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 53
Increasing rate of S$$_N$$1 reaction in the following compounds is:
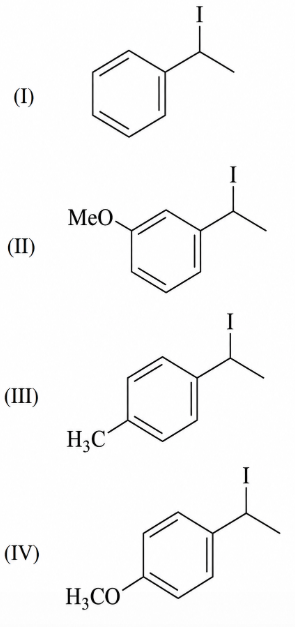
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 54
The major product of the following reaction is:
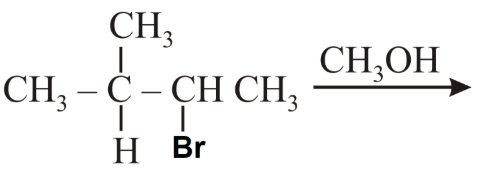
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 55
The major product of the following reaction is:
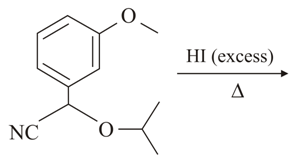
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 56
Major products of the following reaction are:
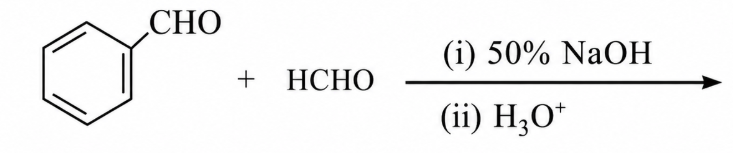
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 57
The major product of the following reaction is:
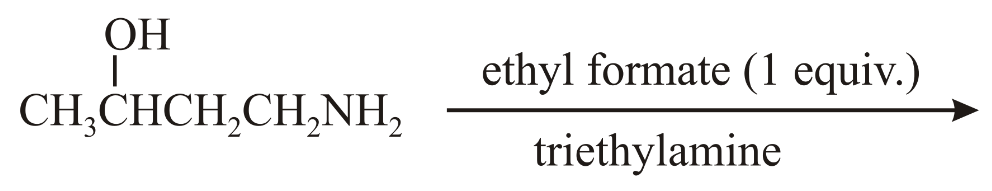
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 58
Ethylamine (C$$_2$$H$$_5$$NH$$_2$$) can be obtained from N-ethylphthalimide on treatment with:
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 59
Which of the following is a condensation polymer?
NTA JEE Main 10th April 2019 Shift 1 - Chemistry - Question 60
Amylopectin is composed of:
.webp)
.webp)
.webp)
.webp)




