Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The increasing order of the reactivity of the following compounds towards electrophilic aromatic substitution reactions is:
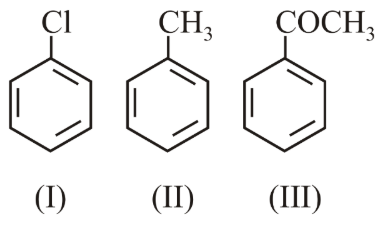
Electrophilic aromatic substitution (EAS) proceeds faster when the benzene ring possesses higher electron density, because the aromatic π-cloud then stabilises the positively-charged σ-complex (arenium ion) that forms in the rate-determining step. In general, an electron-donating group (EDG) activates the ring, while an electron-withdrawing group (EWG) deactivates it. The magnitude of activation or deactivation depends on the balance of two electronic effects:
Inductive effect $$\; (\pm I)$$ and $$\text{Mesomeric / Resonance effect} \; (\pm M)$$
We now examine the substituent already attached to the ring in each of the three given compounds.
For toluene (compound II) the substituent is the methyl group, $$-CH_3$$. A methyl group exhibits a $$+I$$ (electron-releasing inductive) effect and can also donate electron density through hyperconjugation, which behaves like weak resonance donation. Both mechanisms increase electron density on the ring. Therefore toluene is activated toward EAS and reacts faster than benzene.
For chlorobenzene (compound I) the substituent is chlorine. Chlorine is more electronegative than carbon, so it withdraws electron density through the $$-I$$ inductive effect. At the same time, its lone pairs can participate in $$+M$$ resonance donation into the ring. Quantitatively, the $$-I$$ effect of chlorine is stronger than its $$+M$$ effect, giving a net deactivation. However, because some resonance donation is present, the deactivation is only moderate; chlorobenzene reacts a little more slowly than benzene but much faster than rings bearing strongly withdrawing carbonyl groups.
For acetophenone (compound III) the substituent is the acyl group, $$-COCH_3$$. The carbonyl oxygen pulls electron density strongly toward itself by both a powerful $$-M$$ (resonance withdrawal) and a $$-I$$ inductive effect. The combined withdrawal greatly lowers the π-electron density of the ring, making acetophenone strongly deactivated. Its rate toward EAS is far below that of benzene.
Summarising the relative strength of these effects:
$$\text{Rate (EAS)} \;\propto\; \text{Electron density on ring}$$
$$\begin{aligned} -COCH_3 &: \; -M \; \text{(very strong)} + -I \; \Longrightarrow \text{strongly deactivated} \\ Cl &: \; -I \; \text{(strong)} + +M \; \text{(moderate)} \Longrightarrow \text{moderately deactivated} \\ CH_3 &: \; +I \; \text{(moderate)} + \text{hyperconjugation} \Longrightarrow \text{activated} \end{aligned}$$
Hence the rate order is
Acetophenone (III)} \;\lt \; \text{Chlorobenzene (I)} \;\lt \; \text{Toluene (II)
So, the increasing order of reactivity towards electrophilic aromatic substitution is $$\mathrm{III \; \lt \; I \; \lt \; II}$$.
Hence, the correct answer is Option C.

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
