Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
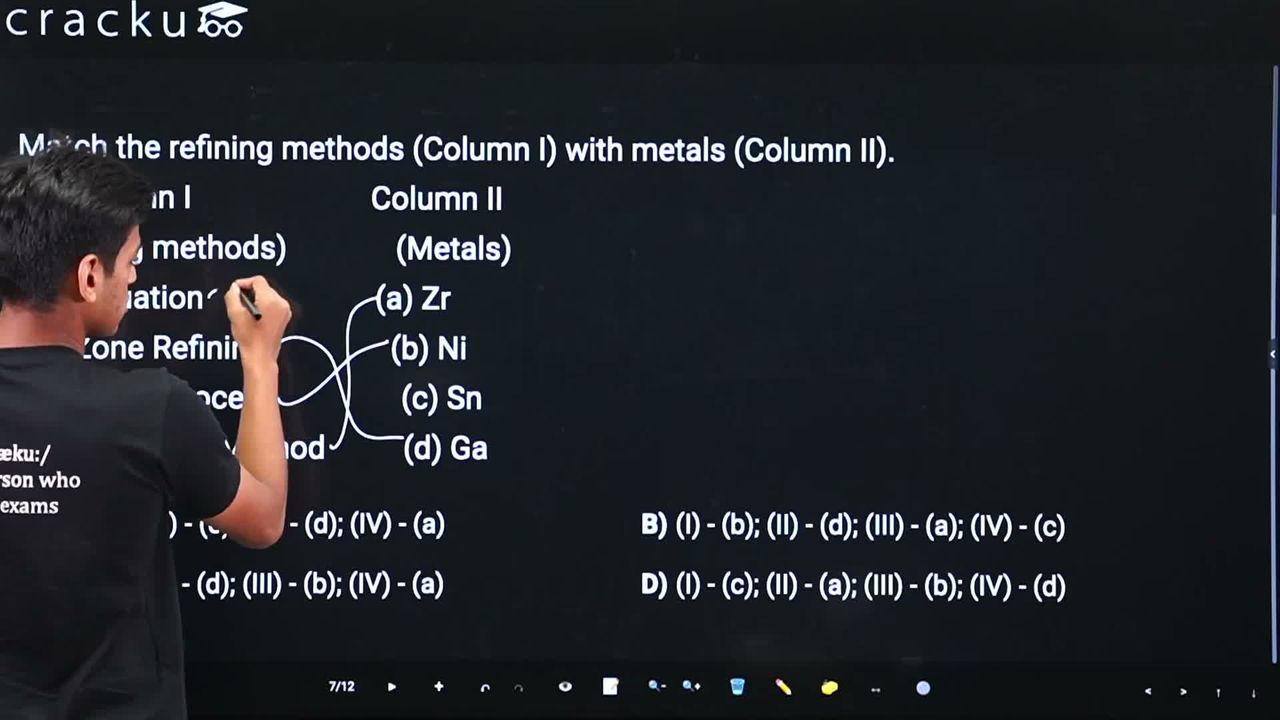
Match the refining methods (Column I) with metals (Column II).
Column I Column II
(Refining methods) (Metals)
(I) Liquation (a) Zr
(II) Zone Refining (b) Ni
(III) Mond Process (c) Sn
(IV) Van Arkel Method (d) Ga
We start by recalling what each refining method is meant to do. Each method is suited to metals that possess particular physical or chemical properties. By matching those properties, we can decide which metal is refined by which method.
First we consider the Liquation process. In Liquation, the impure metal is heated just above its melting point so that the pure metal melts and flows away while the impurities, having higher melting points, remain solid. This method clearly demands a metal with a relatively low melting point. Among the metals listed—$$\text{Zr, Ni, Sn, Ga}$$—we note that $$\text{Sn (tin)}$$ melts at about $$232\ ^\circ\text{C}$$, which is far lower than the melting points of the others. Hence, Sn is refined by Liquation.
Now we look at Zone Refining. Zone Refining is based on the principle that when a molten zone is passed along a solid metal bar, impurities concentrate in the molten region. Repetition moves impurities to one end, leaving ultra-pure metal behind. This technique is wonderfully effective for semiconductors or metals needed in extremely pure form and possessing high tendency to form covalent bonds. Gallium, $$\text{Ga}$$, used in semiconductor devices, is famously purified by this method. So Zone Refining matches Ga.
Next we recall the Mond Process. The Mond Process involves the formation of a volatile metal carbonyl at moderate temperatures followed by its thermal decomposition at higher temperatures to yield pure metal. The key reaction is
$$\text{Ni} + 4\,\text{CO} \;\xrightarrow[50-60^\circ\text{C}]{}\; \text{Ni(CO)}_4$$
followed by
$$\text{Ni(CO)}_4 \;\xrightarrow[180-200^\circ\text{C}]{}\; \text{Ni} + 4\,\text{CO}.$$
This process is characteristic of nickel, $$\text{Ni}$$, because $$\text{Ni(CO)}_4$$ is volatile while the carbonyls of the other listed metals are not similarly convenient. Therefore, the Mond Process is used for Ni.
Finally, we turn to the Van Arkel Method (also called the Van Arkel-de Boer process or the iodide process). It relies on the reversible formation of a volatile metal iodide at moderate temperatures and its subsequent decomposition at a hot filament, depositing the pure metal. The reactions may be written as
$$\text{Zr (impure)} + 2\,\text{I}_2 \;\xrightarrow[500-700^\circ\text{C}]{}\; \text{ZrI}_4$$
and then
$$\text{ZrI}_4 \;\xrightarrow[\,\approx 1800^\circ\text{C}\,]{}\; \text{Zr (pure)} + 2\,\text{I}_2.$$
This technique is especially useful for very reactive transition metals such as zirconium, $$\text{Zr}$$, and titanium. None of the other three metals listed form such stable yet easily decomposed iodides suitable for this method. Thus, the Van Arkel Method corresponds to Zr.
Putting all our matches together, we have
$$\begin{aligned} \text{(I) Liquation} &\;\longrightarrow\; \text{Sn} \; (c),\\ \text{(II) Zone Refining} &\;\longrightarrow\; \text{Ga} \; (d),\\ \text{(III) Mond Process} &\;\longrightarrow\; \text{Ni} \; (b),\\ \text{(IV) Van Arkel Method} &\;\longrightarrow\; \text{Zr} \; (a). \end{aligned}$$
Comparing this sequence with the options provided, we observe that the mapping (I) - (c), (II) - (d), (III) - (b), (IV) - (a) is listed as Option C.
Hence, the correct answer is Option C.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
