NTA JEE Main 9th April 2019 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 31
For a reaction, N$$_2$$(g) + 3H$$_2$$(g) $$\rightarrow$$ 2NH$$_3$$(g), identify di-hydrogen (H$$_2$$) as a limiting reagent in the following reaction mixtures.
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 32
For any given series of spectral lines of atomic hydrogen, let $$\Delta\bar{v} = \bar{v}_{max} - \bar{v}_{min}$$ be the difference in maximum and minimum wave number in cm$$^{-1}$$. The ratio $$\Delta\bar{v}_{Lyman}/\Delta\bar{v}_{Balmer}$$ is:
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 33
The element having the greatest difference between its first and second ionization energies, is:
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 34
Among the following, the molecule expected to be stabilized by anion formation is: C$$_2$$, O$$_2$$, NO, F$$_2$$
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 35
Consider the van der Waal's constants, a and b, for the following gases.

Which gas is expected to have the highest critical temperature?
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 36
Among the following, the set of parameters that represents path functions, is:
i) q + w
ii) q
iii) w
iv) H - TS
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 37
Magnesium powder burns in air to give:
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 38
C$$_{60}$$, an allotrope of carbon contains:
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 39
The correct IUPAC name of the following compound is:
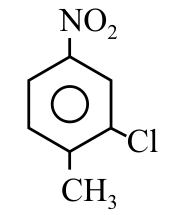
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 40
The increasing order of reactivity of the following compounds towards aromatic electrophilic substitution reaction is:
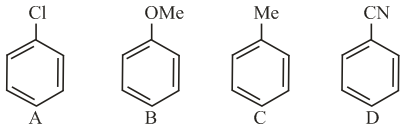
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 41
The major product of the following reaction is:
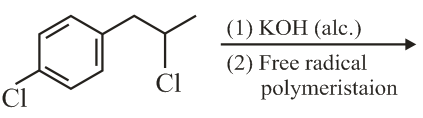
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 42
The major product of the following reaction is:
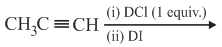
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 43
Excessive release of CO$$_2$$ into the atmosphere results in:
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 44
The osmotic pressure of a dilute solution of an ionic compound XY in water is four times that of a solution of 0.01 M BaCl$$_2$$ in water. Assuming complete dissociation of the given ionic compounds in water, the concentration of XY (in mol L$$^{-1}$$) in solution is:
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 45
Liquid M and liquid N form an ideal solution. The vapour pressures of pure liquids M and N are 450 and 700 mmHg, respectively, at the same temperature. Then correct statements is:
(x$$_M$$ = Mole fraction of 'M' in solution; x$$_N$$ = Mole fraction of 'N' in solution; y$$_M$$ = Mole fraction of 'M' in vapour phase; y$$_N$$ = Mole fraction of 'N' in vapour phase)
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 46
The standard Gibbs energy for the given cell reaction in kJ mol$$^{-1}$$ at 298 K is:
Zn(s) + Cu$$^{2+}$$(aq) $$\rightarrow$$ Zn$$^{2+}$$(aq) + Cu(s),
E$$^0$$ = 2 V at 298 K
(Faraday's constant, F = 96000 C mol$$^{-1}$$)
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 47
The given plots represent the variation of the concentration of a reactant R with time for two different reactions (i) and (ii). The respective orders of the reaction are:
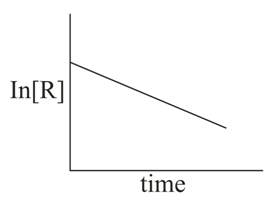
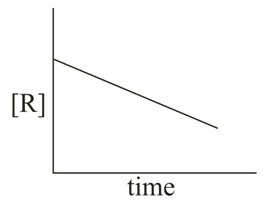
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 48
The aerosol is a kind of colloid in which:
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 49
The ore that contains the metal in the form of fluoride is known as which of the following?
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 50
Match the catalysts (Column I) with products (Column II)
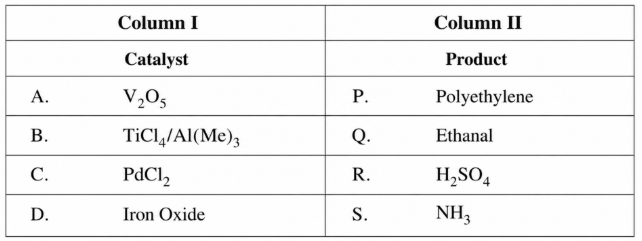
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 51
The correct order of the oxidation states of nitrogen in NO, N$$_2$$O, NO$$_2$$ and N$$_2$$O$$_3$$ is:
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 52
The number of water molecules not coordinated to copper ion directly in CuSO$$_4$$ . 5H$$_2$$O, is:
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 53
The degenerate orbitals of [Cr(H$$_2$$O)$$_6$$]$$^{3+}$$ are:
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 54
The one that will show optical activity is (en = ethane-1, 2-diamine):
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 55
The major product of the following reaction is:
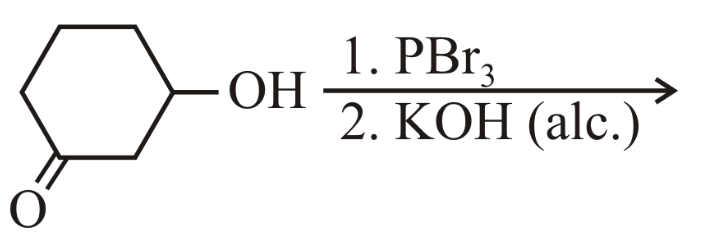
1. PBr$$_3$$
2. KOH (alc.)
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 56
The organic compound that gives following qualitative analysis is:

NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 57
The major product of the following reaction is:
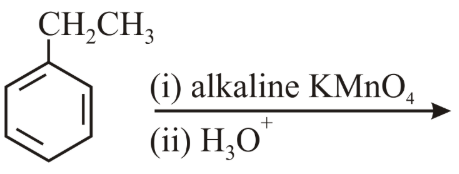
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 58
The major product of the following reaction is:
CH$$_3$$CH = CHCO$$_2$$CH$$_3$$ $$\xrightarrow{\text{LiAlH}_4}$$
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 59
Aniline dissolved in dilute HCl is reacted with sodium nitrite at 0°C. This solution was added dropwise to a solution containing an equimolar mixture of aniline and phenol in dilute HCl. The structure of the major product is:
NTA JEE Main 9th April 2019 Shift 1 - Chemistry - Question 60
Which of the following statements is not true about sucrose?
.webp)
.webp)
.webp)
.webp)




