Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
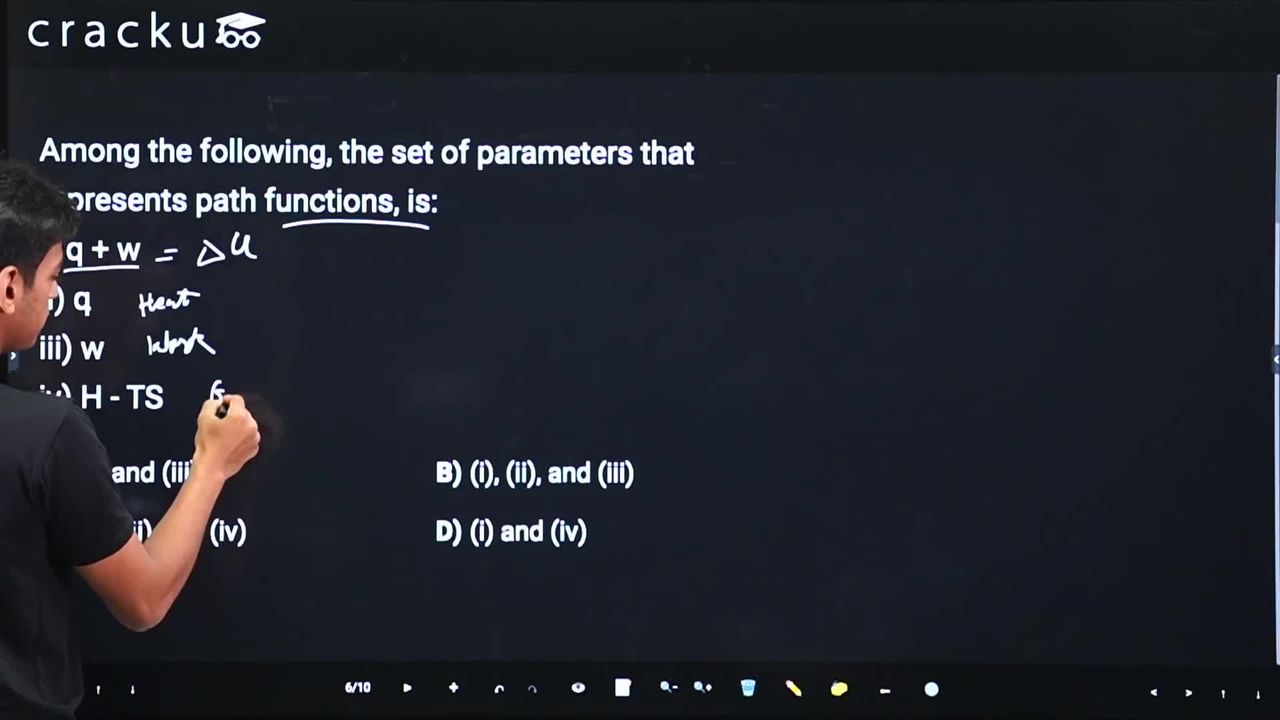
Among the following, the set of parameters that represents path functions, is:
i) q + w
ii) q
iii) w
iv) H - TS
In thermodynamics, we first recall the two fundamental categories of thermodynamic quantities.
• State functions: A quantity that depends only on the current equilibrium state of the system. Its change $$\Delta$$ is independent of the path taken between the initial and final states.
• Path functions: A quantity whose magnitude does depend on the specific path taken during a process. They are written with the differential symbols $$\delta q, \; \delta w$$ to emphasize that they are inexact differentials.
Now we examine each expression in the list one by one.
We have expression (i) $$q + w.$$ From the first law of thermodynamics we state the formula
$$\Delta U = q + w,$$
where $$\Delta U$$ (change in internal energy) is a state function. Because the right-hand side equals a state function, the sum $$q + w$$ behaves as a state function - its value between two fixed states is fixed, no matter which path is taken. Therefore, $$q + w$$ is not a path function.
Next, in expression (ii) we have $$q.$$ Heat $$q$$ transferred between two states varies with the manner (isothermal, adiabatic, reversible, irreversible, etc.) in which the process is carried out. Hence $$q$$ is path dependent. So, $$q$$ is a path function.
In expression (iii) we have $$w.$$ Work $$w$$ done by or on the system also depends on the exact path (for example, different pressure-volume curves give different areas under the curve). Thus $$w$$ is likewise a path function.
Finally, expression (iv) is $$H - TS.$$ Here $$H$$ (enthalpy) and $$S$$ (entropy) are both state functions, while $$T$$ (temperature) is an intensive property that is perfectly defined at equilibrium. Their combination
$$H - TS = G$$
is the Gibbs free energy $$G,$$ which is a well-known state function. Therefore $$H - TS$$ is not a path function.
Summarizing, only $$q$$ and $$w$$ - that is, items (ii) and (iii) - are path functions.
Hence, the correct answer is Option A.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
