Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
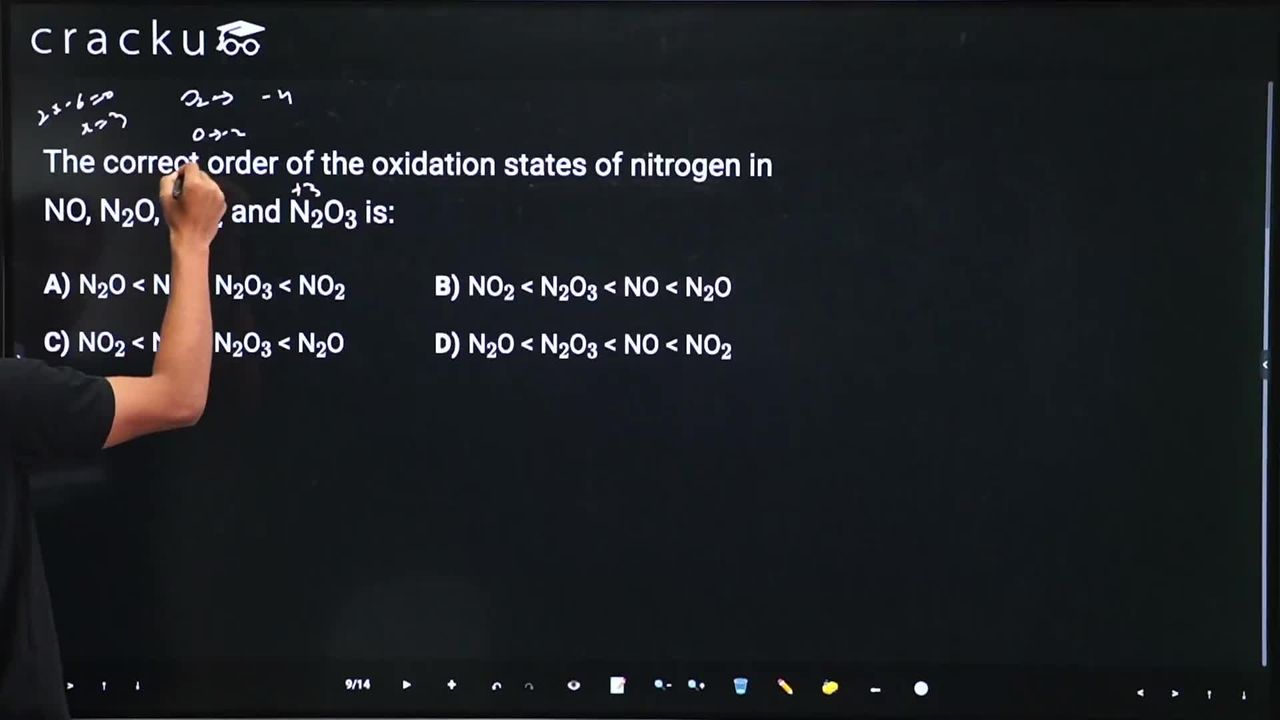
The correct order of the oxidation states of nitrogen in NO, N$$_2$$O, NO$$_2$$ and N$$_2$$O$$_3$$ is:
We recall the general rule that in almost every compound the oxidation state of oxygen is $$-2$$.
For nitric oxide, $$\mathrm{NO}$$: Let the oxidation state of the single nitrogen atom be $$x$$. We have
$$x + (-2) = 0$$
because the molecule is electrically neutral. Solving gives
$$x = +2$$.
For nitrous oxide, $$\mathrm{N_2O}$$: Let each nitrogen atom have the same oxidation state $$x$$. Now
$$2x + (-2) = 0 \quad\Longrightarrow\quad 2x = +2 \quad\Longrightarrow\quad x = +1.$$
For nitrogen dioxide, $$\mathrm{NO_2}$$: The single nitrogen atom has oxidation state $$x$$, so
$$x + 2(-2) = 0 \quad\Longrightarrow\quad x - 4 = 0 \quad\Longrightarrow\quad x = +4.$$
For dinitrogen trioxide, $$\mathrm{N_2O_3}$$: With each nitrogen at oxidation state $$x$$, we write
$$2x + 3(-2) = 0 \quad\Longrightarrow\quad 2x - 6 = 0 \quad\Longrightarrow\quad 2x = +6 \quad\Longrightarrow\quad x = +3.$$
Now we list the values obtained:
$$\mathrm{N_2O}: +1,\qquad \mathrm{NO}: +2,\qquad \mathrm{N_2O_3}: +3,\qquad \mathrm{NO_2}: +4.$$
Arranging them in increasing order we get
$$+1 < +2 < +3 < +4 \;\;\Longrightarrow\;\; \mathrm{N_2O} < \mathrm{NO} < \mathrm{N_2O_3} < \mathrm{NO_2}.$$
Hence, the correct answer is Option A.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
