NTA JEE Main 8th April 2019 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 31
The percentage composition of carbon by mole in methane is:
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 32
0.27 g of a long chain fatty acid was dissolved in 100 cm$$^3$$ of hexane. 10 mL of this solution was added dropwise to the surface of water in a round watch glass. Hexane evaporates and a monolayer is formed. The distance from edge to centre of the watch glass is 10 cm. What is the height of the monolayer?
[Density of fatty acid = 0.9 g cm$$^{-3}$$; $$\pi = 3$$]
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 33
If p is the momentum of the fastest electron ejected from a metal surface after the irradiation of light having wavelength $$\lambda$$, then for 1.5 p momentum of the photoelectron, the wavelength of the light should be: (Assume kinetic energy of ejected photoelectron to be very high in comparison to work function)
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 34
The IUPAC symbol for the element with atomic number 119 would be:
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 35
Among the following molecules/ions, $$C_2^{2-}$$, $$N_2^{2-}$$, $$O_2^{2-}$$, $$O_2$$, which one is diamagnetic and has the shortest bond length?
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 36
5 moles of an ideal gas at 100 K are allowed to undergo reversible compression till its temperature becomes 200 K. If $$C_V = 28$$ J K$$^{-1}$$, calculate $$\Delta U$$ and $$\Delta pV$$ for the process. (R = 8.0 J K$$^{-1}$$ mol$$^{-1}$$)
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 37
For the following reaction, equilibrium constants are given:
$$S(s) + O_2(g) \rightleftharpoons SO_2(g); \quad K_1 = 10^{52}$$
$$2S(s) + 3O_2(g) \rightleftharpoons 2SO_3(g); \quad K_2 = 10^{129}$$
The equilibrium constant for the reaction, $$2SO_2(g) + O_2(g) \rightleftharpoons 2SO_3(g)$$ is:
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 38
The strength of 11.2 volume solution of H$$_2$$O$$_2$$ is:
[Given that, the molar mass of H = 1 g mol$$^{-1}$$ and O = 16 g mol$$^{-1}$$]
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 39
The covalent alkaline earth metal halide X = Cl, Br, I is:
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 40
Which of the following compounds will show the maximum 'enol' content?
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 41
Polysubstitution is a major drawback in:
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 42
Which one of the following alkenes when treated with HCl yields majorly an anti Markovnikov product?
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 43
The maximum prescribed concentration of copper in drinking water is:
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 44
Consider the bcc unit cells of the solids 1 and 2 with the position of atoms as shown below. The radius of atom B is twice that of atom A. The unit cell edge length is 50% more in solid 2 than in 1. What is the approximate packing efficiency in solid 2?
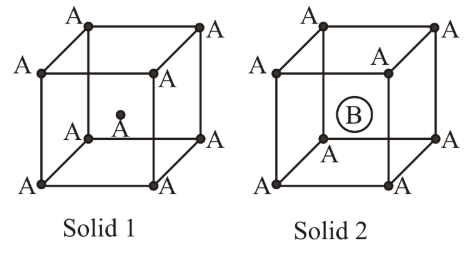
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 45
For the solution of the gases w, x, y and z in water at 298 K, the Henry's law constants (K$$_H$$) are 0.5, 2, 35 and 40 kbar, respectively. The correct plot for the given data is:
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 46
Calculate the standard cell potential (in V) of the cell in which the following reaction takes place:
Fe$$^{2+}$$(aq) + Ag$$^+$$(aq) $$\rightarrow$$ Fe$$^{3+}$$(aq) + Ag(s)
Given that $$E^{0}_{Ag^+/Ag} = x$$ V, $$E^{0}_{Fe^{2+}/Fe} = y$$ V, $$E^{0}_{Fe^{3+}/Fe} = z$$ V
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 47
For a reaction scheme A $$\xrightarrow{k_1}$$ B $$\xrightarrow{k_2}$$ C, if the net rate of formation of B is set to be zero then the concentration of B is given by:
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 48
The Mond process is used for the:
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 49
The ion that has sp$$^3$$d$$^2$$ hybridization for the central atom is:
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 50
The correct statement about ICl$$_5$$ and ICl$$_4^{-}$$ is:
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 51
The statement that is INCORRECT about the interstitial compounds is:
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 52
The compound that inhibits the growth of tumors is:
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 53
The calculated spin-only magnetic moments BM of the anionic and cationic species of FeH$$_2$$O$$_6^{+2}$$ and Fe(CN)$$_6^{4-}$$ respectively, are:
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 54
The major product of the following reaction is:
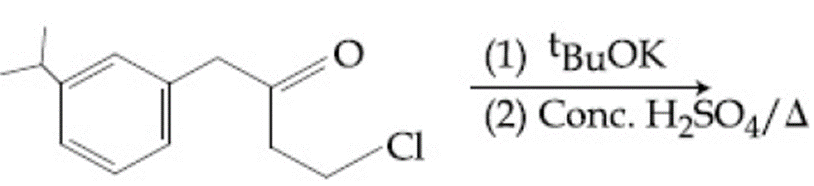
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 55
The major product of the following reaction is:
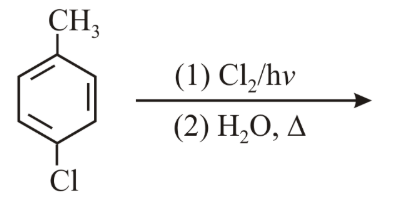
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 56
The major product obtained in the following reaction is:
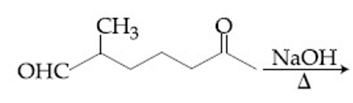
NaOH, $$\Delta$$
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 57
The major product obtained in the following reaction is:
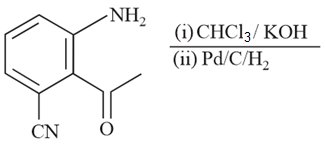
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 58
The major product in the following reaction is:
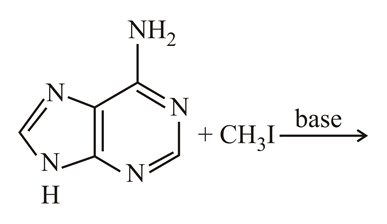
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 59
The structure of Nylon-6 is:
NTA JEE Main 8th April 2019 Shift 2 - Chemistry - Question 60
Fructose and glucose can be distinguished by:
.webp)
.webp)
.webp)
.webp)




