Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
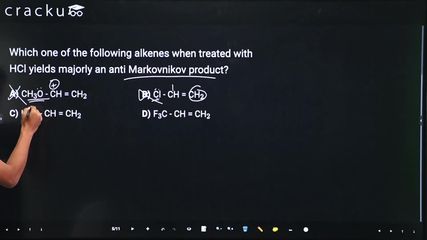
Which one of the following alkenes when treated with HCl yields majorly an anti Markovnikov product?
First we recall the general statement of Markovnikov’s rule: on electrophilic addition of $$\mathrm{HX}$$ to an unsymmetrical alkene, the proton $$\left(\mathrm{H^+}\right)$$ attaches to that double-bonded carbon which already bears the larger number of hydrogen atoms, because this mode of attack generates the more stable carbocation. Put algebraically, if the alkene is $$\mathrm{C_a=C_b}$$, then in the Markovnikov pathway $$\mathrm{H^+}$$ goes to the carbon rich in hydrogens and the intermediate carbocation appears on the other carbon.
The stability of the intermediate carbocation is decided mainly by two electronic effects: the inductive effect $$\left(-I\ \text{or}\ +I\right)$$ and the mesomeric or resonance effect $$\left(-M\ \text{or}\ +M\right)$$ of the substituent already attached to that carbon. A group which donates electron density by $$+I$$ or $$+M$$ stabilises the positive charge, whereas a group which withdraws electron density by a strong $$-I$$ effect destabilises it. Hence, if a substituent has a very powerful $$-I$$ effect and lacks any meaningful $$+M$$ donation, the carbocation situated adjacent to it becomes highly unstable, and the reaction may proceed by the opposite orientation, i.e. the anti-Markovnikov mode.
Now we analyse each given alkene one by one.
Option A : $$\mathrm{CH_3O{-}CH=CH_2}$$
The methoxy group $$\left(\mathrm{-OCH_3}\right)$$ exerts a $$+M$$ resonance donation because of the lone pairs on oxygen. In the Markovnikov pathway the proton adds to the terminal $$\mathrm{CH_2}$$ carbon and the carbocation appears on the carbon bearing $$\mathrm{-OCH_3}$$:
$$\mathrm{CH_3O{-}CH=CH_2\ \overset{H^+}{\underset{\text{Markovnikov}}{\rightarrow}}\ CH_3O{-}\overset{+}{\mathrm{CH}}{-}CH_3$$
The positive charge is strongly stabilised by resonance with the oxygen lone pair, so this path is preferred. Hence the product is Markovnikov, not anti-Markovnikov.
Option B : $$\mathrm{Cl{-}CH=CH_2}$$
Chlorine possesses a weak $$+M$$ effect as well as a $$-I$$ effect. When the carbocation is placed on the chlorine-bearing carbon (Markovnikov orientation) the lone pairs of chlorine can offer some resonance stabilisation:
$$\mathrm{Cl{-}CH=CH_2\ \overset{H^+}{\underset{\text{Markovnikov}}{\rightarrow}}\ Cl{-}\overset{+}{\mathrm{CH}}{-}CH_3}$$
The $$+M$$ relief outweighs the inductive withdrawal; consequently the Markovnikov product remains major. No anti-Markovnikov preference is observed.
Option C : $$\mathrm{H_2N{-}CH=CH_2}$$
The amino group $$\left(\mathrm{-NH_2}\right)$$ is a very strong $$+M$$ donor. Exactly as in Option A, the Markovnikov pathway places the positive charge adjacent to the donor nitrogen, which is highly stabilising by resonance:
$$\mathrm{H_2N{-}CH=CH_2\ \overset{H^+}{\underset{\text{Markovnikov}}{\rightarrow}}\ H_2N{-}\overset{+}{\mathrm{CH}}{-}CH_3}$$
Therefore this alkene also obeys Markovnikov’s rule.
Option D : $$\mathrm{CF_3{-}CH=CH_2}$$
The trifluoromethyl group $$\left(\mathrm{-CF_3}\right)$$ is one of the strongest electron-withdrawing groups known; it shows an intense $$-I$$ effect and has no $$+M$$ resonance donation, because the fluorines are not conjugated with the carbocation centre through a lone-pair bridge as chlorine can be. Examine the two possible orientations:
Markovnikov attack (proton to the terminal $$\mathrm{CH_2}$$ carbon):
$$\mathrm{CF_3{-}CH=CH_2\ \overset{H^+}{\underset{\text{Markovnikov}}{\rightarrow}}\ CF_3{-}\overset{+}{\mathrm{CH}}{-}CH_3}$$
The carbocation $$\mathrm{CF_3{-}\overset{+}{CH}{-}CH_3}$$ is directly attached to the very powerful $$-I$$ group $$\mathrm{CF_3}$$, so the positive charge is pulled even further electron-deficient and becomes extremely unstable.
Anti-Markovnikov attack (proton to the $$\mathrm{CF_3{-}CH}$$ carbon):
$$\mathrm{CF_3{-}CH=CH_2\ \overset{H^+}{\underset{\text{anti-M}}{\rightarrow}}\ CF_3{-}CH_2{-}\overset{+}{\mathrm{CH}}_2$$
Now the positive charge resides on the terminal carbon, which is not adjacent to the $$\mathrm{-CF_3}$$ group, so it does not suffer the strong inductive withdrawal. Even though both carbocations are primary, the one formed in the anti-Markovnikov pathway is appreciably more stable because it is farther from the electron-withdrawing group. Therefore the reaction proceeds predominantly by this orientation, giving the anti-Markovnikov product $$\mathrm{CF_3{-}CH_2{-}CH_2Cl}$$.
Thus, among the four alkenes, only the trifluoromethyl-substituted alkene in Option D furnishes the anti-Markovnikov addition product with $$\mathrm{HCl}$$.
Hence, the correct answer is Option D.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
