NTA JEE Main 9th April 2019 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 31
What would be the molality of 20% (mass/mass) aqueous solution of KI? (molar mass of KI = 166 g mol$$^{-1}$$)
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 32
Which one of the following about an electron occupying the 1s orbital in a hydrogen atom is incorrect? (The Bohr radius is represented by a$$_0$$).
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 33
Among the following species, the diamagnetic molecule is:
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 34
At a given temperature T, gases Ne, Ar, Xe and Kr are found to deviate from ideal gas behaviour. Their equation of state is given as $$p = \frac{RT}{V - b}$$ at T. Here, b is the van der Waals constant. Which gas will exhibit steepest increase in the plot of Z (compression factor) vs p?
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 35
During compression of a spring the work done is 10 kJ and 2 kJ escaped to the surroundings as heat. The change in internal energy, $$\Delta U$$ (in kJ) is:
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 36
In an acid-base titration, 0.1 M HCl solution was added to the NaOH solution of unknown strength. Which of the following correctly shows the change of pH of the titration mixture in this experiment?
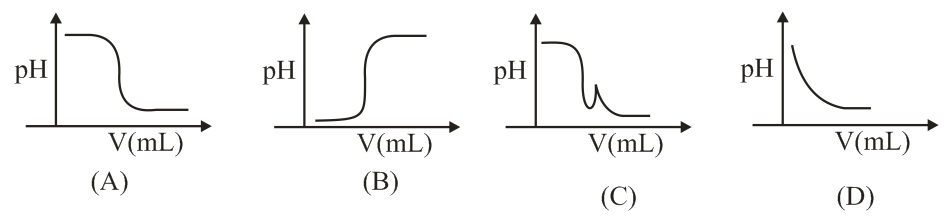
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 37
The structures of beryllium chloride in the solid state and vapour phase, respectively, are:
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 38
The correct statements among I to III regarding group 13 element oxides are,
(I) Boron trioxide is acidic.
(II) Oxides of aluminum and gallium are amphoteric.
(III) Oxides of indium and thallium are basic.
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 39
The amorphous form of silica is:
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 40
The layer of atmosphere between 10 km to 50 km above the sea level is called as:
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 41
Molal depression constant for a solvent is 4.0 K kg mol$$^{-1}$$. The depression in the freezing point of the solvent for 0.03 mol kg$$^{-1}$$ solution of K$$_2$$SO$$_4$$ is: (Assume complete dissociation of the electrolyte)
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 42
A solution of Ni(NO$$_3$$)$$_2$$ is electrolyzed between platinum electrode 0.1 Faraday electricity. How many mole of Ni will be deposited at the cathode?
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 43
Consider the given plot of enthalpy of the following reaction between A and B.
A + B $$\rightarrow$$ C + D. Identify the incorrect statement.
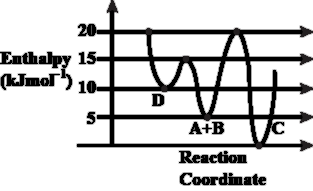
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 44
10 mL of 1 mM surfactant solution forms a monolayer covering 0.24 cm$$^2$$ on a polar substrate. If the polar head is approximated as a cube, what is its edge length?
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 45
The one that is not a carbonate ore is:
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 46
Assertion: For the extraction of iron, haematite ore is used.
Reason: Haematite is a carbonate ore of iron.
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 47
HF has highest boiling point among hydrogen halides, because it has:
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 48
The maximum number of possible oxidation states of actinoids are shown by:
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 49
The maximum possible denticities of a ligand given below towards a common transition and inner-transition metal ion, respectively, are:
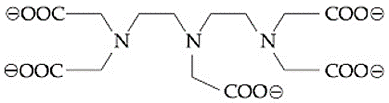
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 50
The correct statements among I to III are:
(I) Valence bond theory cannot explain the color exhibited by transition metal complexes.
(II) Valence bond theory can predict quantitatively the magnetic properties of transition metal complexes.
(III) Valence bond theory cannot distinguish ligands as weak and strong field ones.
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 51
Increasing order of reactivity of the following compounds for S$$_N$$1 substitution is:
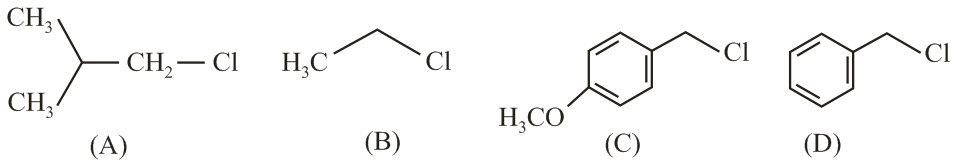
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 52
Which of the following potential energy (P.E.) diagrams represents the S$$_N$$1 reaction?
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 53
p-Hydroxybenzophenone upon reaction with bromine in carbon tetrachloride gives:
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 54
In the following reaction:
carbonyl compound + MeOH $$\overset{\mathrm{HCl}}{\rightleftharpoons}$$ acetal
Rate of the reaction is the highest for:
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 55
The major product of the following reaction is:
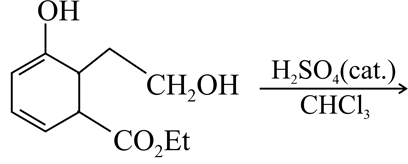
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 56
The major products A and B for the following reactions are, respectively:
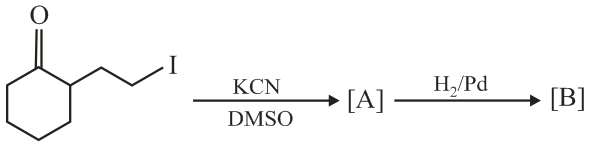
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 58
Which of the following compounds is a constituent of the polymer
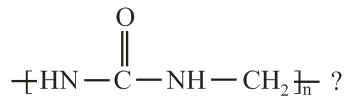
?
NTA JEE Main 9th April 2019 Shift 2 - Chemistry - Question 60
The peptide that gives positive ceric ammonium nitrate and carbylamine tests is:
.webp)
.webp)
.webp)
.webp)




