NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 51
From the following, the least stable structure is :
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 52
Which of the following represents the correct trend for the mentioned property ?
A.$$F > P > S > B$$ - First Ionization Energy
B.$$Cl > F > S > P$$ - Electron Affinity
C. $$K > Al > Mg > B$$ - Metallic character
D. $$K_{2}O > Na_{2}O > MgO > AL_{2}O_{3}$$ - Basic character
Choose the correct answer from the options given below :
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 53
80 mL of a hydrocarbon on mixing with 264 mL of oxygen in a closed U-tube undergoes complete combustion. The residual gases after cooling to 273 K occupy 224 mL. When the system is treated with KOH solution, the volume decreases to 64 ml. The formula of the hydrocarbon is:
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 54
Given below are two statements:
Statement I: Among $$[Cu(NH_{3})_{4}]^{2+},[Ni(en)_{3})]^{2+},[Ni(NH_{3})_{6}]^{2+}$$ and $$[Mn(H_{2}O)_{6}]^{2+},[Mn(H_{2}O)_{6}]^{2+}$$ has the maximum number of unpaired electrons.
Statement II : The number of pairs among $$\left\{[Ni(Cl_{4}]^{2-},[Ni(CO)_{4}]\right\}$$, $$\left\{[NiCl_{4}]^{2-},[Ni(CN)_{4}]^{2-}\right\}$$ and $$\left\{[Ni(CO)_{4}],[Ni(CN)_{4}]^{2-}\right\}$$ that contain only diamagnetic species is two.
ln the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 55
Identify the correct statements.
A. Arginine and Tryptophan are essential amino acids.
B. Histidine does not contain heterocyclic ring in its structure.
C. Proline is a six membered cyclic ring amino acid.
D. Glycine does not have chiral centre.
E. Cysteine has characteristic feature of side chain as $$MeS-CH_{2}-CH_{2}-.$$
CHoose the correct answer from the options given below :
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 56
Consider the following reactions.
$$PbCl_{2}+K_{2}CrO_{4}\rightarrow A+2KCI$$
(Hot solution)
$$A+NaOH\rightleftharpoons B+Na_{2}CrO_{4}$$
$$PbSO_{4}+4CH_{3}COONH_{4}\rightarrow (NH_{4})_{2}SO_{4}+X$$
In the above reactions, A, Band X are respectively.
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 57
$$\text{MnO}_4^{2-}$$, in acidic medium, disproportionates to :
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 58
Given below are two statements:
Statement I : When an electric discharge is passed through gaseous hydrogen, the hydrogen molecules dissociate and the energetically excited hydrogen atoms produce electromagnetic radiation of discrete frequencies.
Statement II: The frequency of second line of Balmer series obtained from He+ is equal to that of first line of Lyman series obtained from hydrogen a tom.
ln the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 59
An organic compound "P" of molecular formula $$C_6H_{12}O_3$$ gives positive Iodoform test but negative Tollen's test. When "P" is treated with dilute acid, it produces "Q". "Q" gives positive Tollen's test and also iodoform test. The structure of "P" is :
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 60
14.0 g of calcium metal is allowed to react with excess HCI at 1.0 atm pressure and 273 K
Which of the following statements is incorrect?
[Given : Molar mass in g $$\text{mol}^{-1}$$ of Ca-40, Cl-35.5, H-1]
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 61
Given below are two statements:
Statement I: The number of pairs among $$[SiO_{2},CO_{2}],[SnO,SnO_{2}],[PbO,PbO_{2]}$$ and $$[GeO,GeO_{2}]$$, which contain oxides that are both amphoteric is 2.
Statement ll: $$BF_{3}$$ is an electron deficient molecule, can act as a Lewis add, forms adduct with $$NH_{3}$$ and has a trigonal planar geometry.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 62
Which of the following graphs between pressure 'p' versus volume 'V' represents the maximum work done?
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 63
Identify A in the following reaction.
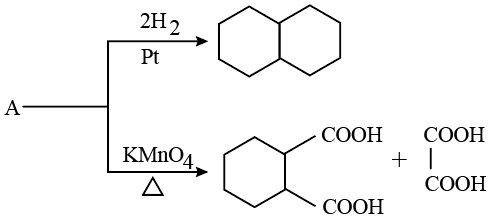
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 64
A hydrocarbon 'P' $$(C_{4}H_{8})$$ on reaction with HCl gives an optically active compound 'Cl' $$(C_{4}H_{9}Cl)$$ which on reaction with one mole of ammonia gives compound 'R' $$(C_{4}H_{11}N)$$ on diazolization followed by hydrolysis gives 'S'. Identify P, Q, Rand S.
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 65
Elements P and Q form two types of non-volatile, non-ionizable compounds PQ and $$PQ_{2}$$. When 1g of PQ is dissolved in 50 g of solvent ''A', $$\Delta T_{b}$$was 1.176 K while when 1 g of $$PQ_{2}$$ is dissolved in 50g of solvent 'A'.$$\Delta T_{b}$$ was 0.689 K ($$K_{b}$$ of 'A' =5K kg $$mol^{-1}$$) The molar masses of elements P and Q (in g $$mol^{-1}$$ ) respectively, are:
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 66
Identify correct statements from the following :
A Propanal and propanone are functional isomers.
B. Ethoxyethane and methoxypropane are metamers.
C. But-2-ene shows optical isomerism.
D. But-1-ene and but-2-ene are functional isomers.
E. Pentane and 2, 2-dimethyl propane are chain isomers.
Choose the correct answer from the options given below:
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 67
An organic compound (P) on treatment with aqueous ammonia under hot condition forms compound (Q) which on heating with $$Br_{2}$$ and KOH forms compound (R) having molecular formula $$C_{6}H_{7}N$$ Names of P, Q and R respectively are.
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 68
Given below are two statements:
Statement I :
The number of species among $$SF_{4},NH_4^+,[NiCl_{4}]^{2-},XeF_{4},[PtCl_{4}]^{2-},SeF_{4}$$ and $$[Ni(CN)_{4}]^{2-}$$, tha t have tetrahedral geometry is 3.
Statement II :
In the set $$[NO_{2},BeH_{2},BF_{3},AlCl_{3}]$$ all the molecules have incomplete octet around central atom.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 69
In Carius method, 0.75 g of an organic compound gave 1.2 g of barium sulphate, find percentage of sulphur (molar mass 32 g $$mol^{-1}$$ ) Molar mass of barium sulphate is 233 g $$mol^{-1}$$
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 70
For the reaction $$N_{2}O_{4}\rightleftharpoons2NO_{2}$$ , graph is plotted as shown below. Identify correct statements.
A. Standard free energy change for the reaction is $$-5.40kJmol^{-1}$$.
B. As $$\triangle G^{\ominus}$$ in graph is positive, $$N_{2}O_{4}$$ will not dissociate into $$NO_{2}$$ at all.
C. Reverse reaction will go to completion.
D. When 1 mole of $$N_{2}O_{4}$$ changes into equilibrium mixture, value of $$\triangle G^{\ominus}$$ = -0.84kJ $$mol^{-1}$$.
E. When 2 mole of $$NO_{2}$$ changes into equilibrium mixture, $$\triangle G^{\ominus}$$ for equilibrium mixture is -6.24kJ $$mol^{-1}$$.
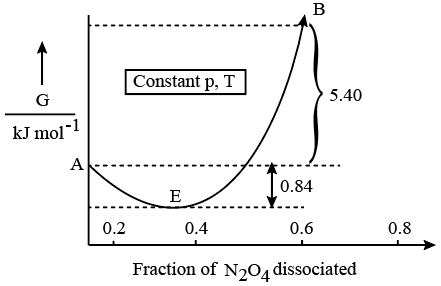
Choose the correct answer from the options given below:
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 71
Consider the following reaction sequence
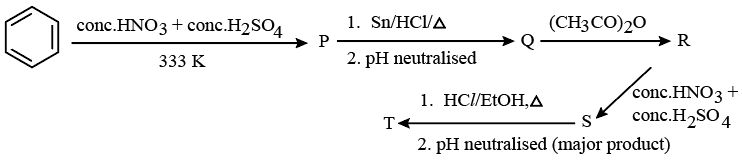
The percentage of nitrogen in product 'T' formed is ____ %. (Nearest integer)
(Given molar mass in g $$mol^{-1}$$ H : 1, C: 12, N : 14, 0: 16)
789
456
123
0.-
Clear All
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 72
Pre-exponential factors of two different reactions of same order are identical. Let activation energy of first reaction exceeds the activation energy of second reaction by 20 kJ $$mol^{-1}$$. If $$k_{1}\text{ and }k_{2}$$ are the rate constants of first and second reaction respectively at 300 K, then In $$\frac{k_{2}}{k_{1}}$$ will be ___.
(nearest integer) $$[R=8.3JK^{-1}mol^{-1}]$$
789
456
123
0.-
Clear All
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 73
Use the following data :
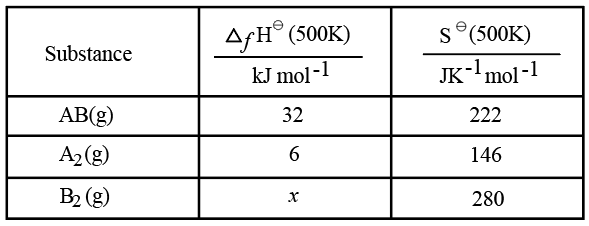
One mole each of $$A_{2}(g)$$ and $$B_{2}(g)$$ are taken in a 1 L closed flask and allowed to establish the equilibrium at 500K
$$A_{2}(g)+B_{2}(g)\rightleftharpoons2AB(g)$$
The value of x in $$( kJ mol^{-1})$$ is ____ . (Nearest integer)
(Given: log K=2.2 R= 8.3 kJ $$K^{-1} mol^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 74
Consider the following reactions:
$$NaCl+K_{2}Cr_{2}O_{7}+H_{2}SO_{4}\rightarrow A+KHSO_{4}+NaHSO_{4}+H_{2}O$$
$$A+NaOH\rightarrow B+NaCl+H_{2}O$$
$$B+H_{2}SO_{4}+H_{2}O_{2}\rightarrow C+Na_{2}SO_{4}+H_{2}O$$
In the product 'C, 'X' is the number of $$O_{2}^{2-}$$ units, 'Y' is the total number oxygen atoms present and 'Z' is the oxidation state of Cr·. The value of X + Y + Z is ______
789
456
123
0.-
Clear All
NTA JEE Mains 21st Jan 2026 Shift 1 - Chemistry - Question 75
The pH and conductance of a weak acid (HX) was found to be 5 and $$4\times10^{-5}S$$. respectively. The conductance was measured under standard condition using a cell where the electrode plates having a surface area of 1 $$cm^{2}$$ were at a distance of 15 cm apart. The value of the limiting molar conductivity is ______ S $$m^{2}mol^{-1}$$ (nearest integer)
(Given : degree of dissociation of the weak acid ($$\alpha$$) < < 1)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




