Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
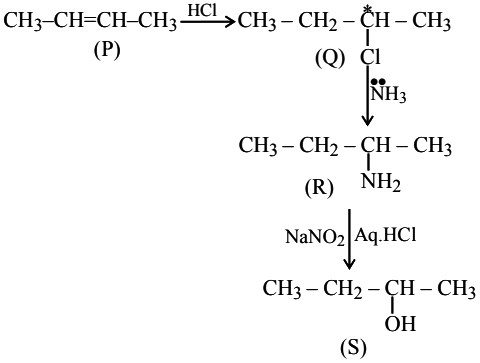
A hydrocarbon 'P' $$(C_{4}H_{8})$$ on reaction with HCl gives an optically active compound 'Cl' $$(C_{4}H_{9}Cl)$$ which on reaction with one mole of ammonia gives compound 'R' $$(C_{4}H_{11}N)$$ on diazolization followed by hydrolysis gives 'S'. Identify P, Q, Rand S.

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
