Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
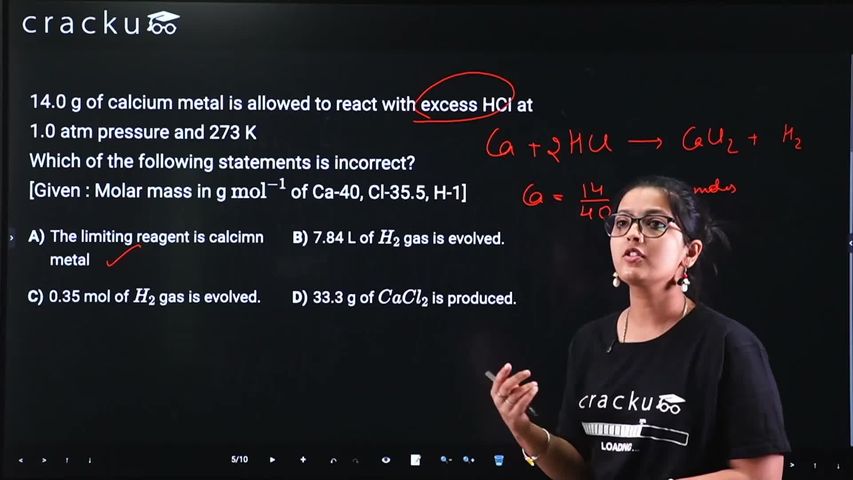
14.0 g of calcium metal is allowed to react with excess HCI at 1.0 atm pressure and 273 K
Which of the following statements is incorrect?
[Given : Molar mass in g $$\text{mol}^{-1}$$ of Ca-40, Cl-35.5, H-1]
Ca + 2HCl → CaCl₂ + H₂. Moles Ca = 14/40 = 0.35 mol.
Moles H₂ = 0.35 mol. Volume at STP = 0.35 × 22.4 = 7.84 L ✓.
Mass CaCl₂ = 0.35 × 111 = 38.85 g. Option 4 says 33.3 g — incorrect.
The answer is Option 4: 33.3 g of CaCl₂ is the incorrect statement.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
