NTA JEE Main 12th April 2019 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 31
25 g of an unknown hydrocarbon upon burning produces 88 g of CO$$_2$$ and 9 g of H$$_2$$O. This unknown hydrocarbon contains:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 32
Among the following, the energy of 2s orbital is lowest in:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 33
In comparison to boron, beryllium has:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 34
The incorrect match in the following is:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 35
In which one of the following equilibria, K$$_p$$ ≠ K$$_c$$?
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 36
The molar solubility of Cd(OH)$$_2$$ is $$1.84 \times 10^{-5}$$ M in water. The expected solubility of Cd(OH)$$_2$$ in a buffer solution of pH = 12 is:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 37
The temporary hardness of a water sample is due to compound X. Boiling this sample converts X to compound Y. X and Y, respectively, are:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 38
The incorrect statement is:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 39
The C - C bond length is maximum in:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 40
The IUPAC name for the following compound is:
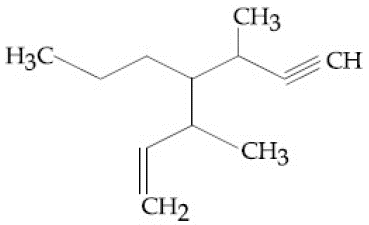
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 41
Which one of the following is likely to give a precipitate with AgNO$$_3$$ solution?
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 42
In the following skew conformation of ethane, H' - C - C - H'' dihedral angle is:
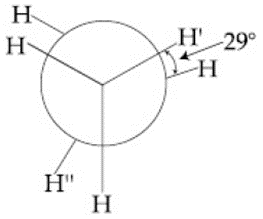
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 43
Heating of 2-chloro-1-phenylbutane with EtOK/EtOH gives X as the major product. Reaction X with Hg(OAc)$$_2$$/H$$_2$$O followed by NaBH$$_4$$ gives Y as the major product. Y is:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 44
The primary pollutant that leads to photochemical smog is:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 45
The ratio of number of atoms present in a simple cubic, body centered cubic and face centered cubic structure are, respectively:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 46
A solution is prepared by dissolving 0.6 g of urea (molar mass = 60 g mol$$^{-1}$$) and 1.8 g of glucose (molar mass = 180 g mol$$^{-1}$$) in 100 mL of water at 27°C. The osmotic pressure of the solution is:
(R = 0.08206 L atm K$$^{-1}$$ mol$$^{-1}$$)
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 47
The decreasing order of electrical conductivity of the following aqueous solutions is:
(A) 0.1 M Formic acid,
(B) 0.1 M Acetic acid,
(C) 0.1 M Benzoic acid.
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 48
NO$$_2$$ required for a reaction is produced by the decomposition of N$$_2$$O$$_5$$ in CCl$$_4$$ as per the equation, 2N$$_2$$O$$_5$$(g) $$\to$$ 4NO$$_2$$(g) + O$$_2$$(g). The initial concentration of N$$_2$$O$$_5$$ is 3.00 mol L$$^{-1}$$ and it is 2.75 mol L$$^{-1}$$ after 30 minutes. The rate of formation of NO$$_2$$ is:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 49
Among the following, the incorrect statement about colloids is:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 50
The correct statement is:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 51
The pair that has similar atomic radii is:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 52
Thermal decomposition of a Mn compound (X) at 513 K results in compound Y, MnO$$_2$$ and a gaseous product. MnO$$_2$$ reacts with NaCl and concentrated H$$_2$$SO$$_4$$ to give a pungent gas Z. X, Y and Z, respectively, are:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 53
The coordination numbers of Co and Al in [Co(Cl)(en)$$_2$$]Cl and K$$_3$$[Al(C$$_2$$O$$_4$$)$$_3$$], respectively are:
(en = ethane-1, 2-diamine)
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 54
The compound used in the treatment of lead poisoning is:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 55
An 'Assertion' and a 'Reason' is given below. Choose the correct answer from the following options:
Assertion (A): Vinyl halides do not undergo nucleophilic substitution easily.
Reason (R): Even though the intermediate carbocation is stabilized by loosely held $$\pi$$-electrons, the cleavage is difficult because of the strong bonding.
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 56
Consider the following reactions:
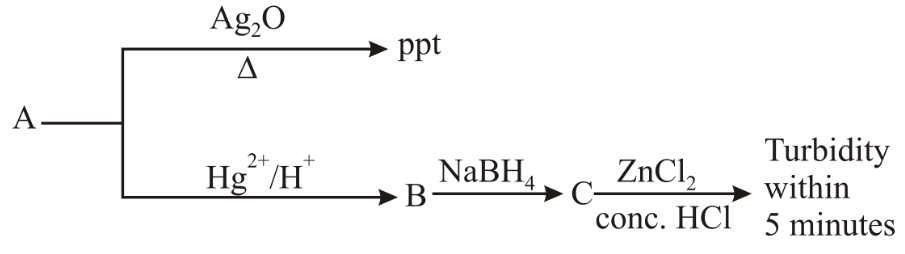
'A' is:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 57
What will be the major product when m-cresol is reacted with propargyl bromide (HC ≡ C - CH$$_2$$Br) in presence of K$$_2$$CO$$_3$$ in acetone?
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 58
Benzene diazonium chloride on reaction with aniline in the presence of dilute hydrochloric acid gives:
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 59
The correct name of the following polymer is:
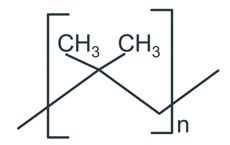
NTA JEE Main 12th April 2019 Shift 2 - Chemistry - Question 60
Which of the given statements is incorrect about glycogen?
.webp)
.webp)
.webp)
.webp)




