Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
For every gaseous equilibrium we recall the relation
$$K_p=K_c\;(RT)^{\Delta n_g}$$
where $$\Delta n_g=\left(\text{total moles of gaseous products}\right)-\left(\text{total moles of gaseous reactants}\right).$$
If $$\Delta n_g=0$$, the factor $$(RT)^{\Delta n_g}=(RT)^0=1$$ and we immediately obtain $$K_p=K_c$$.
If $$\Delta n_g\neq 0$$, then $$(RT)^{\Delta n_g}\neq 1$$ and we have $$K_p\neq K_c$$. So our task reduces to evaluating $$\Delta n_g$$ for each given equilibrium.
Option A : $$2\,\text{HI}(g)\;\rightleftharpoons\;\text{H}_2(g)+\text{I}_2(g)$$
Total gaseous reactant moles $$=2$$, product moles $$=1+1=2$$. Hence
$$\Delta n_g = 2-2 = 0 \Longrightarrow K_p=K_c.$$
Option B : $$2\,\text{C}(s)+\text{O}_2(g)\;\rightleftharpoons\;2\,\text{CO}(g)$$
Only gases are counted, so solid carbon is ignored. Reactant gaseous moles $$=1$$, product gaseous moles $$=2$$. Therefore
$$\Delta n_g = 2-1 = +1 \neq 0 \Longrightarrow K_p\neq K_c.$$
Option C : $$2\,\text{NO}(g)\;\rightleftharpoons\;\text{N}_2(g)+\text{O}_2(g)$$
Reactant moles $$=2$$, product moles $$=1+1=2$$. Thus
$$\Delta n_g = 2-2 = 0 \Longrightarrow K_p=K_c.$$
Option D : $$\text{NO}_2(g)+\text{SO}_2(g)\;\rightleftharpoons\;\text{NO}(g)+\text{SO}_3(g)$$
Reactant moles $$=1+1=2$$, product moles $$=1+1=2$$. Hence
$$\Delta n_g = 2-2 = 0 \Longrightarrow K_p=K_c.$$
Among the four equilibria, only Option B gives $$\Delta n_g\neq 0$$ and therefore satisfies $$K_p\neq K_c$$.
Hence, the correct answer is Option B.
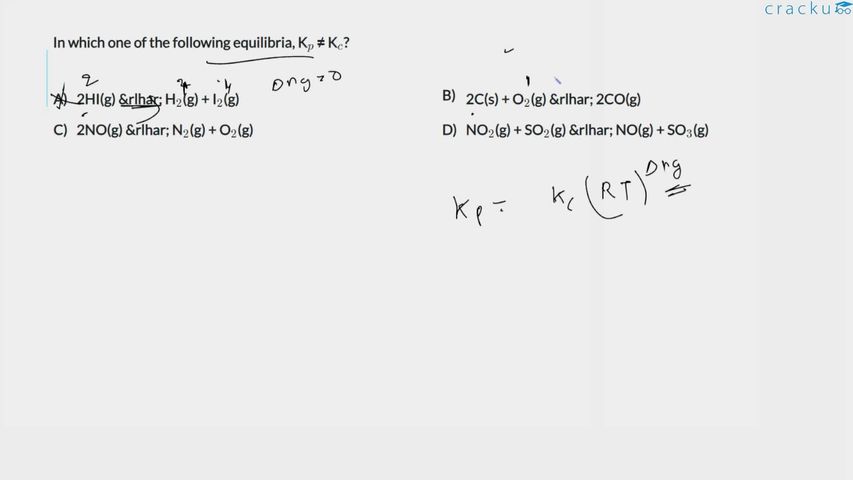
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
