Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The ratio of number of atoms present in a simple cubic, body centered cubic and face centered cubic structure are, respectively:
First, we recall that a unit cell can have atoms at its corners, at the centres of its faces and/or at its body centre. Whenever we count the effective number of atoms inside one unit cell, we must take into account the sharing of these positions with neighbouring cells.
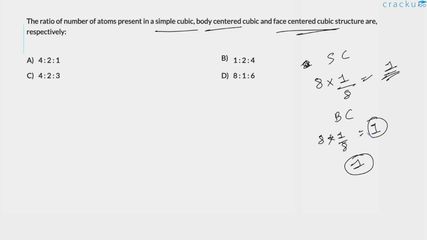
Corner contribution. A corner atom is shared by 8 adjacent unit cells. Hence each corner atom contributes
$$\text{Contribution of one corner atom}= \dfrac{1}{8}$$
There are always 8 corners in a cubic unit cell, so the total contribution coming from all the corner atoms is
$$8\times\dfrac{1}{8}=1$$
Now we examine the three specific types of cubic lattices one by one.
1. Simple Cubic (SC). The simple cubic unit cell has atoms only at the 8 corners and nowhere else. Therefore the effective number of atoms per simple-cubic unit cell is
$$N_{\text{SC}} = 1$$
2. Body-centred Cubic (BCC). Besides the 8 corner atoms already counted, a BCC unit cell possesses one additional atom at its body centre. The body-centre atom is not shared with any other unit cell, so it contributes fully:
$$\text{Body-centre contribution}=1$$
Hence,
$$N_{\text{BCC}} = 1\;(\text{corners}) + 1\;(\text{body centre}) = 2$$
3. Face-centred Cubic (FCC). In an FCC cell, in addition to the 8 corner atoms we have one atom at the centre of each of the 6 faces. A face-centre atom is shared by 2 adjacent unit cells, thus each face-centre atom contributes
$$\dfrac{1}{2}$$
With 6 faces the total face-centre contribution is
$$6 \times \dfrac{1}{2} = 3$$
Therefore,
$$N_{\text{FCC}} = 1\;(\text{corners}) + 3\;(\text{faces}) = 4$$
We have now found the number of atoms per unit cell for all three lattices:
$$N_{\text{SC}} : N_{\text{BCC}} : N_{\text{FCC}} = 1 : 2 : 4$$
Hence, the correct answer is Option B.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
