Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Benzene diazonium chloride on reaction with aniline in the presence of dilute hydrochloric acid gives:
We are asked to predict the product when benzene diazonium chloride reacts with aniline in the presence of dilute hydrochloric acid. First, let us write the formulas of the reactants in a compact mathematical form.
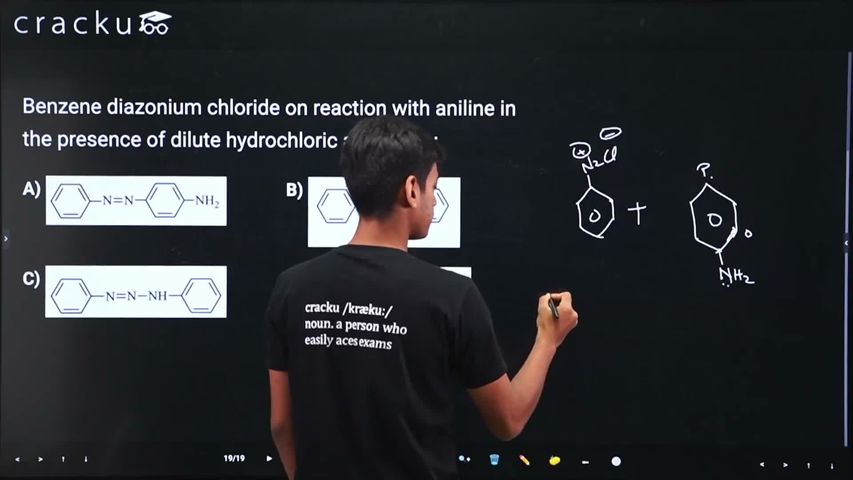
Benzene diazonium chloride is represented as $$\text{C}_6\text{H}_5{-}\overset{+}{\text{N}}\!\!=\!\!\text{N}\; \text{Cl}^-$$ while aniline is $$\text{C}_6\text{H}_5\text{NH}_2$$
In the presence of dilute $$\text{HCl}$$, a small fraction of aniline gets protonated to the anilinium ion $$\text{C}_6\text{H}_5\text{NH}_3^{+}\;,$$ but a sufficient amount of the free -NH2 form is still present because the acid is only dilute. The unprotonated aniline ring therefore behaves as an activated aromatic system; its electron-releasing $$-\,\text{NH}_2$$ group makes the ortho and para positions strongly nucleophilic toward an electrophile.
The diazonium cation $$\text{C}_6\text{H}_5\!-\!{\overset{+}{\text N}}\!\!=\!\!\text N$$ is a very good electrophile. Hence the well-known azo-coupling reaction (an electrophilic aromatic substitution) occurs. The coupling takes place preferentially at the para site of aniline because the ortho sites are sterically hindered by the -NH2 group. Writing the step explicitly, we have
$$ \text{C}_6\text{H}_5{-}\overset{+}{\text{N}}\!\!=\!\!\text{N}\; \text{Cl}^- \;+\; \text{C}_6\text{H}_4\big(\text{NH}_2\big)\; \longrightarrow\; \text{C}_6\text{H}_5{-}\text{N}{=}\text{N}{-}\text{C}_6\text{H}_4\big(\text{NH}_2\big)_{p} \;+\; \text{Cl}^- . $$
The product contains an -N=N- azo linkage bridging two benzene rings, and the -NH2 group of the second ring occupies the para position with respect to that bridge. The systematic name of this dye is p-aminoazobenzene; it is also called aniline yellow.
Because an -N=N- linkage (not an -N=N-NH- linkage) is formed, the compound is an azo compound, not a diazoamino compound. Among the given choices, only Option A correctly describes this structure.
Hence, the correct answer is Option A.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
