NTA JEE Main 12th April 2019 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 31
5 moles of AB$$_2$$ weigh $$125 \times 10^{-3}$$ kg and 10 moles of A$$_2$$B$$_2$$ weigh $$300 \times 10^{-3}$$ kg. The molar mass of A (M$$_A$$) in kg mol$$^{-1}$$ are:
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 32
The electrons are more likely to be found:
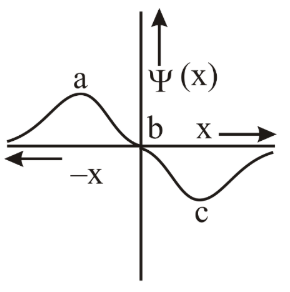
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 33
The group number, number of valence electrons, and valency of an element with atomic number 15, respectively, are:
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 34
The correct statement among the following is:
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 35
An ideal gas is allowed to expand from 1 L to 10 L against a constant external pressure of 1 bar. The work done in kJ is:
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 36
Enthalpy of sublimation of iodine is 24 cal g$$^{-1}$$ at 200°C. If specific heat of I$$_2$$(s) and I$$_2$$(vap) are 0.055 and 0.031 cal g$$^{-1}$$ K$$^{-1}$$ respectively, then enthalpy of sublimation of iodine at 250°C in cal g$$^{-1}$$ is:
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 37
What is the molar solubility of Al(OH)$$_3$$ in 0.2 M NaOH solution? Given that, solubility product of Al(OH)$$_3$$ = $$2.4 \times 10^{-24}$$:
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 38
An example of a disproportionation reaction is
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 39
Given:
Co$$^{3+}$$ + e$$^-$$ $$\to$$ Co$$^{2+}$$; E° = +1.81V
Pb$$^{3+}$$ + 2e$$^-$$ $$\to$$ Pb$$^{2+}$$; E° = +1.67V
Ce$$^{4+}$$ + e$$^-$$ $$\to$$ Ce$$^{3+}$$; E° = +1.61V
Bi$$^{3+}$$ + 3e$$^-$$ $$\to$$ Bi; E° = +0.20V
Oxidizing power of the species will increase in the order:
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 40
The correct sequence of thermal stability of the following carbonates is:
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 41
The basic structural unit of feldspar, zeolites, mica, and asbestos is:
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 42
The increasing order of the pK$$_b$$ of the following compound is:
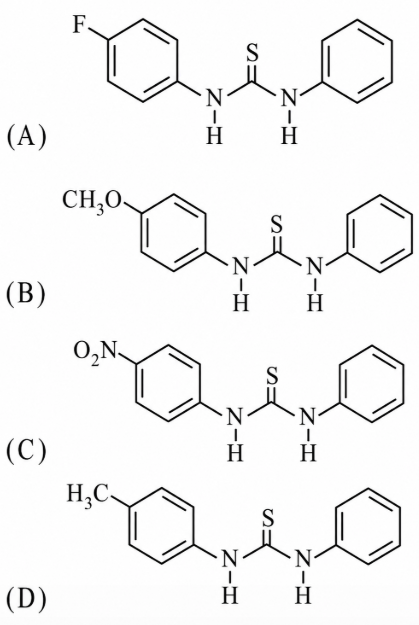
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 43
The major product(s) obtained in the following reaction is/are:
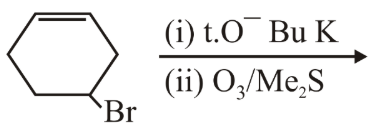
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 44
The major product of the following addition reaction is
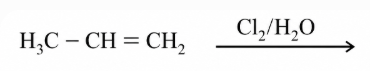
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 45
But-2-ene on reaction with alkaline KMnO$$_4$$ at elevated temperature followed by acidification will give:
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 46
The correct set of species responsible for the photochemical smog is:
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 47
An element has a face-centered cubic fcc structure with a cell edge of a. The distance between the centres of two nearest tetrahedral voids in the lattice is
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 48
The mole fraction of a solvent in aqueous solution of a solute is 0.8. The molality (in mol kg$$^{-1}$$) of the aqueous solution is:
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 49
In the following reaction; xA $$\to$$ yB
$$\log_{10}\left(-\frac{dA}{dt}\right) = \log_{10}\left(-\frac{dB}{dt}\right) + 0.3010$$
'A' and 'B' respectively can be:
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 50
Peptization is a:
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 51
The idea of froth flotation method came from a person X and this method is related to the process Y of ores. X and Y, respectively, are:
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 52
The metal that gives hydrogen gas upon treatment with both the acid as well as the base is
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 53
The complex ion that will lose its crystal field stabilization energy upon oxidation of metal to +3 state is:
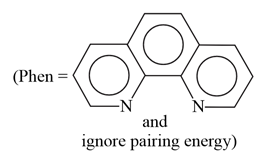
(Phen = phenanthroline, ignore pairing energy)
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 54
Complete removal of both the axial ligands (along the z-axis) from an octahedral complex leads to which of the following splitting patterns? (relative orbital energies not on scale).
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 55
The major products of the following reaction are:
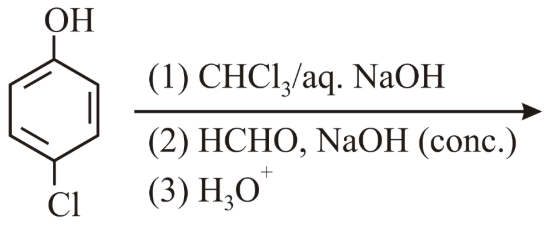
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 56
The major product of the following reaction:
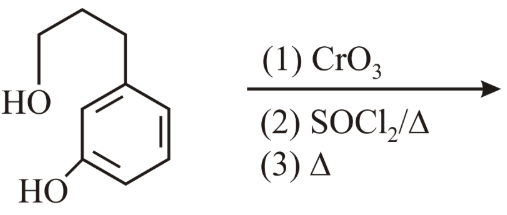
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 57
Which of the following is a thermosetting polymer?
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 58
An organic compound 'A' is oxidized with Na$$_2$$O$$_2$$ followed by boiling with HNO$$_3$$. The resultant solution is then treated with ammonium molybdate to yield a yellow precipitate. Based on above observation, the element present in the given compound is:
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 59
Glucose and Galactose are having identical configuration in all the positions except position.
NTA JEE Main 12th April 2019 Shift 1 - Chemistry - Question 60
Which of the following statements is not true about RNA?
.webp)
.webp)
.webp)
.webp)




