NTA JEE Mains 8th April 2024 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 31
Identify the correct statements about p-block elements and their compounds. (A) Non metals have higher electronegativity than metals. (B) Non metals have lower ionisation enthalpy than metals. (C) Compounds formed between highly reactive nonmetals and highly reactive metals are generally ionic. (D) The non-metal oxides are generally basic in nature. (E) The metal oxides are generally acidic or neutral in nature. Choose the correct answer from the options given below :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 32
The shape of carbocation is :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 33
When $$\psi_A$$ and $$\psi_B$$ are the wave functions of atomic orbitals, then $$\sigma^*$$ is represented by :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 34
The equilibrium $$Cr_2O_7^{2-} \rightleftharpoons 2CrO_4^{2-}$$ is shifted to the right in :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 35
Given below are two statements : Statement (I) : A Buffer solution is the mixture of a salt and an acid or a base mixed in any particular quantities. Statement (II) : Blood is naturally occurring buffer solution whose pH is maintained by $$H_2CO_3/HCO_3^-$$ concentrations. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 36
The correct sequence of acidic strength of the following aliphatic acids in their decreasing order is: $$CH_3CH_2COOH$$, $$CH_3COOH$$, $$CH_3CH_2CH_2COOH$$, $$HCOOH$$
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 37
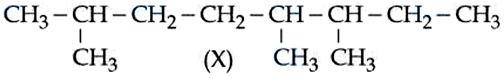
IUPAC name of following hydrocarbon (X) is :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 38
Given below are two statements : Statement (I) : Kjeldahl method is applicable to estimate nitrogen in pyridine. Statement (II) : The nitrogen present in pyridine can easily be converted into ammonium sulphate in Kjeldahl method. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 39
In qualitative test for identification of presence of phosphorous, the compound is heated with an oxidising agent. Which is further treated with nitric acid and ammonium molybdate respectively. The yellow coloured precipitate obtained is :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 40
The emf of cell
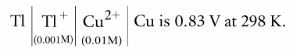
It could be increased by :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 41
The reaction; $$\frac{1}{2}H_{2(g)} + AgCl_{(s)} \rightarrow H^+_{(aq)} + Cl^-_{(aq)} + Ag_{(s)}$$ occurs in which of the following galvanic cell :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 42
For a reaction $$A \xrightarrow{K_1} B \xrightarrow{K_2} C$$. If the rate of formation of $$B$$ is set to be zero then the concentration of $$B$$ is given by :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 43
Identify the incorrect statements about group 15 elements : (A) Dinitrogen is a diatomic gas which acts like an inert gas at room temperature. (B) The common oxidation states of these elements are $$-3, +3$$ and $$+5$$. (C) Nitrogen has unique ability to form $$p\pi - p\pi$$ multiple bonds. (D) The stability of $$+5$$ oxidation states increases down the group. (E) Nitrogen shows a maximum covalency of 6. Choose the correct answer from the options given below :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 44
Given below are two statements : Statement (I) : Fusion of $$MnO_2$$ with KOH and an oxidising agent gives dark green $$K_2MnO_4$$. Statement (II) : Manganate ion on electrolytic oxidation in alkaline medium gives permanganate ion. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 45
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 46
Given below are two statements : Statement (I) : $$S_N2$$ reactions are 'stereospecific', indicating that they result in the formation of only one stereo-isomer as the product. Statement (II) : $$S_N1$$ reactions generally result in formation of product as racemic mixtures. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 47
Which one of the following compounds will readily react with dilute NaOH?
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 48
Match List - I with List - II.
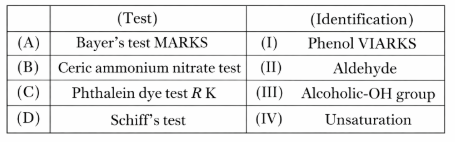
Choose the correct answer from the options given below :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 49
Match List - I with List - II.
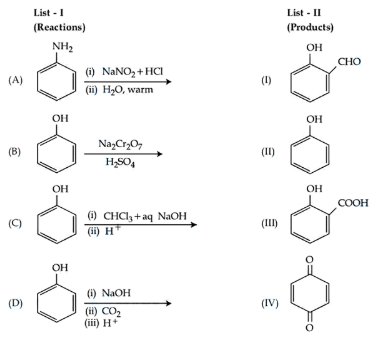
Choose the correct answer from the options given below :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 50
Given below are two statements : Statement (I) : All the following compounds react with p-toluenesulfonyl chloride. $$C_6H_5NH_2$$, $$(C_6H_5)_2NH$$, $$(C_6H_5)_3N$$. Statement (II) : Their products in the above reaction are soluble in aqueous NaOH. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 51
Wavenumber for a radiation having wavelength $$5800 \text{ Å}$$ is $$x \times 10 \text{ cm}^{-1}$$. The value of $$x$$ is _____.
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 52
Number of molecules having bond order 2 from the following molecules is _____ : $$C_2, O_2, Be_2, Li_2, Ne_2, N_2, He_2$$
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 53
$$\Delta_{vap}H^{\ominus}$$ for water is $$+40.79 \text{ kJ mol}^{-1}$$ at 1 bar and $$100°C$$. Change in internal energy for this vapourisation under same condition is _____ $$\text{kJ mol}^{-1}$$. (Given $$R = 8.3 \text{ JK}^{-1}\text{mol}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 54
Total number of optically active compounds from the following is ____
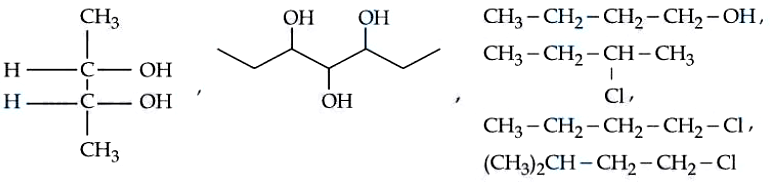
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 55
Total number of aromatic compounds among the following compounds is _____
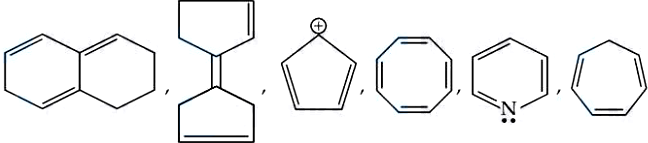
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 56
A solution is prepared by adding 1 mole ethyl alcohol in 9 mole water. The mass percent of solute in the solution is _____ (Given : Molar mass in g/mol: Ethyl alcohol : 46, water: 18)
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 57
Molality of an aqueous solution of urea is 4.44 m. Mole fraction of urea in solution is $$x \times 10^{-3}$$. Value of $$x$$ is _____.
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 58
Total number of unpaired electrons in the complex ions $$[Co(NH_3)_6]^{3+}$$ and $$[NiCl_4]^{2-}$$ is _____
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 59
Two moles of benzaldehyde and one mole of acetone under alkaline conditions using aqueous NaOH after heating gives $$x$$ as the major product. The number of $$\pi$$ bonds in the product $$x$$ is _____
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 2 - Chemistry - Question 60
The total number of carbon atoms present in tyrosine, an amino acid, is _____
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




