Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
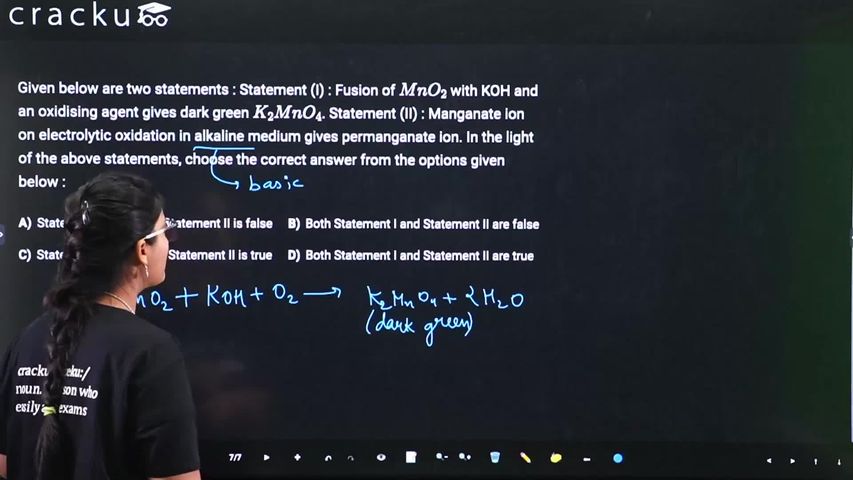
Given below are two statements : Statement (I) : Fusion of $$MnO_2$$ with KOH and an oxidising agent gives dark green $$K_2MnO_4$$. Statement (II) : Manganate ion on electrolytic oxidation in alkaline medium gives permanganate ion. In the light of the above statements, choose the correct answer from the options given below :
We need to evaluate both statements about manganese compounds.
First, fusion of $$MnO_2$$ with KOH and an oxidising agent gives dark green $$K_2MnO_4$$ by a standard preparation method for potassium manganate.
$$2MnO_2 + 4KOH + O_2 \rightarrow 2K_2MnO_4 + 2H_2O$$
Here, $$MnO_2$$ (Mn in +4 state) is oxidised to $$K_2MnO_4$$ (Mn in +6 state) and the product is indeed dark green in colour. Therefore, the first statement is true.
Next, the manganate ion $$MnO_4^{2-}$$ can be oxidised to the permanganate ion $$MnO_4^{-}$$ by electrolytic oxidation in an alkaline solution.
$$MnO_4^{2-} \rightarrow MnO_4^{-} + e^{-}$$
This is a well-known method for preparing $$KMnO_4$$ from $$K_2MnO_4$$, so the second statement is also true.
Since both statements are true, the correct answer is Option 4: Both Statement I and Statement II are true.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
