Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Total number of optically active compounds from the following is ____
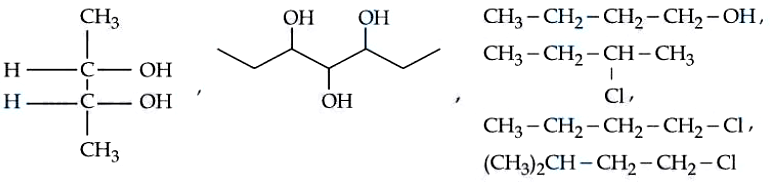
Correct Answer: 1
CH3-CH(OH)-CH(OH)-CH3: This Fischer projection shows chiral centers at C2 and C3. However, there is a horizontal plane of symmetry passing directly between C2 and C3, dividing the molecule into two identical halves. This makes it a meso compound, optically inactive.
Heptane-3,4,5-triol: The molecule has chiral centers at C3 and C5, and a pseudoasymmetric center at C4. As drawn, there is a vertical plane of symmetry passing straight through C4 and its attached -OH group. The left side (ethyl group and -OH group) perfectly mirrors the right side. Thus, it is a meso compound.
Butanol: There are no chiral carbons (no carbon is attached to four different groups). Hence optically inactive.
1-chlorobutane: There are no chiral carbons. Hence Optically inactive.
1-chloro-3-methylbutane: There are no chiral carbons. Carbon-3 is attached to two identical methyl groups. Hence, optically inactive.
2-chlorobutane: Carbon-2 is bonded to four distinct groups. This makes C2 a chiral center. There is no plane or center of symmetry in the molecule. Hence optically active compound.
Total Optically active compounds = 0+0+0+0+0+1 = 1

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
