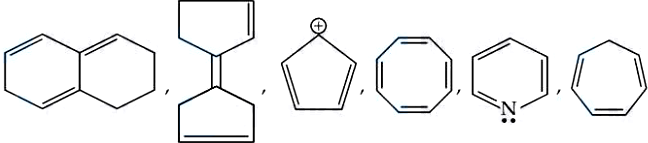
To determine if a molecule is aromatic, it must satisfy Hückel's Rule. A compound is aromatic if it is:
- Cyclic
- Planar
- Fully Conjugated (has a p-orbital on every atom in the ring)
- Contains (4n + 2) pi electrons (where n is an integer: 2, 6, 10, 14...).
Bicyclic Polyene: This is not fully conjugated throughout the entire system. One of the rings has sp3 hybridized carbons (saturated carbons), which breaks the conjugation. Non-aromatic.
Fulvalene derivative: While it has conjugated double bonds, the cross-conjugated double bond between the rings doesn't create a single cyclic path of (4n+2) electrons. Non-aromatic.
Cyclopentadienyl Cation: This molecule is cyclic, planar, and fully conjugated, but it only has 4pi electrons. This follows the 4n rule. Anti-aromatic.
Cyclooctatetraene (COT): This has 8pi electrons. According to the 4n rule, it should be anti-aromatic, but to avoid instability, the molecule adopts a "tub" shape (non-planar).
Non-aromatic.
Pyridine: The nitrogen atom is sp^2 hybridized. The lone pair on the nitrogen is in an sp^2 orbital perpendicular to the p-orbitals, so it does not count toward the pi system. The ring has 6pi electrons (n=1). Aromatic.
Cycloheptatriene: This molecule has a CH2 group (sp^3 hybridized) in the ring. This breaks the continuous overlap of p-orbitals. Non-aromatic.