Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
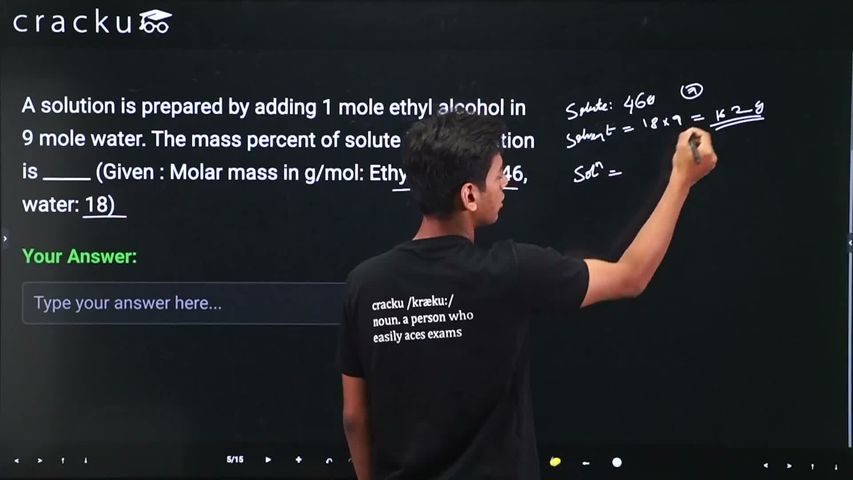
A solution is prepared by adding 1 mole ethyl alcohol in 9 mole water. The mass percent of solute in the solution is _____ (Given : Molar mass in g/mol: Ethyl alcohol : 46, water: 18)
Correct Answer: 22
We need to find the mass percent of ethyl alcohol in a solution containing 1 mole ethyl alcohol and 9 moles of water.
Since the mass percent of solute is given by $$\text{Mass percent} = \frac{\text{Mass of solute}}{\text{Mass of solution}} \times 100$$, we note that the molar mass of ethyl alcohol is 46 g/mol and that of water is 18 g/mol.
Now the mass of ethyl alcohol in the solution is $$1 \times 46 = 46$$ g, and the mass of water is $$9 \times 18 = 162$$ g. Substituting these values, the total mass of the solution becomes $$46 + 162 = 208$$ g.
Therefore the mass percent of ethyl alcohol is $$\text{Mass percent} = \frac{46}{208} \times 100 = \frac{4600}{208} = 22.115\%$$, which rounds to approximately $$22\%$$.
The answer is $$\boxed{22}$$.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
