Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The equilibrium $$Cr_2O_7^{2-} \rightleftharpoons 2CrO_4^{2-}$$ is shifted to the right in :
We need to determine in which medium the equilibrium $$Cr_2O_7^{2-} \rightleftharpoons 2CrO_4^{2-}$$ shifts to the right.
Write the complete ionic equilibrium
The interconversion between dichromate and chromate ions in aqueous solution involves $$H^+$$ and $$OH^-$$ ions. The complete equation is:
$$Cr_2O_7^{2-} + 2OH^- \rightleftharpoons 2CrO_4^{2-} + H_2O$$
Alternatively, in terms of $$H^+$$:
$$2CrO_4^{2-} + 2H^+ \rightleftharpoons Cr_2O_7^{2-} + H_2O$$
Apply Le Chatelier's Principle
The forward reaction (dichromate to chromate) consumes $$OH^-$$ ions. According to Le Chatelier's Principle:
- In a basic medium (excess $$OH^-$$): The high concentration of $$OH^-$$ drives the equilibrium to the right, favouring the formation of yellow $$CrO_4^{2-}$$ (chromate) ions.
- In an acidic medium (excess $$H^+$$): The $$OH^-$$ concentration is low, so the equilibrium shifts to the left, favouring the orange $$Cr_2O_7^{2-}$$ (dichromate) ions.
Conclusion
This is why dichromate solutions appear orange in acidic conditions and turn yellow (chromate) in basic conditions. The equilibrium shifts to the right in a basic medium.
The correct answer is Option (2): a basic medium.
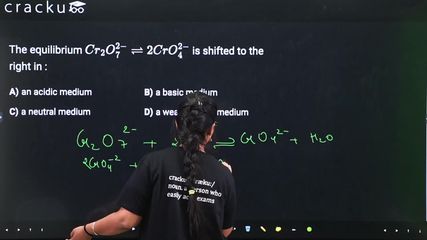
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
