NTA JEE Mains 8th April 2024 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 31
Combustion of glucose $$(C_6H_{12}O_6)$$ produces $$CO_2$$ and water. The amount of oxygen (in g) required for the complete combustion of $$900 \text{ g}$$ of glucose is : [Molar mass of glucose in $$\text{gmol}^{-1} = 180$$]
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 32
Match List I with List II
Choose the correct answer from the options given below:
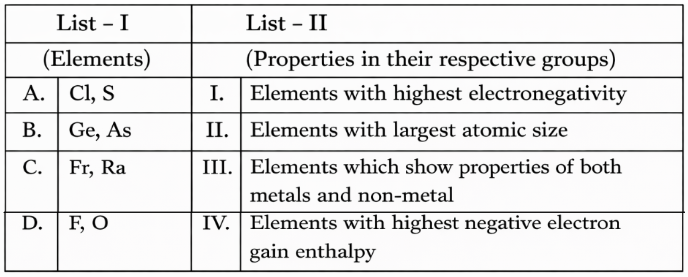
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 33
Match List I with List II
Choose the correct answer from the options given below:
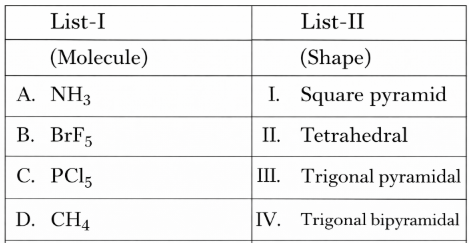
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 34
Match List I with List II
Choose the correct answer from the options given below:
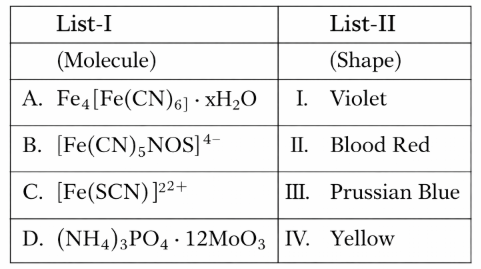
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 35
Given below are two statements: Statement I: $$N(CH_3)_3$$ and $$P(CH_3)_3$$ can act as ligands to form transition metal complexes. Statement II: As N and P are from same group, the nature of bonding of $$N(CH_3)_3$$ and $$P(CH_3)_3$$ is always same with transition metals. In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 36
For the given hypothetical reactions, the equilibrium constants are as follows: $$X \rightleftharpoons Y; K_1 = 1.0$$, $$Y \rightleftharpoons Z; K_2 = 2.0$$, $$Z \rightleftharpoons W; K_3 = 4.0$$. The equilibrium constant for the reaction $$X \rightleftharpoons W$$ is
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 37
Among the following halogens $$F_2, Cl_2, Br_2$$ and $$I_2$$. Which can undergo disproportionation reactions?
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 38
Thiosulphate reacts differently with iodine and bromine in the reactions given below: $$2S_2O_3^{2-} + I_2 \rightarrow S_4O_6^{2-} + 2I^-$$, $$S_2O_3^{2-} + 5Br_2 + 5H_2O \rightarrow 2SO_4^{2-} + 4Br^- + 10H^+$$. Which of the following statement justifies the above dual behaviour of thiosulphate?
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 39
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R: Assertion A: The stability order of +1 oxidation state of Ga, In and Tl is $$Ga < In < Tl$$. Reason R: The inert pair effect stabilizes the lower oxidation state down the group. In the light of the above statements, choose the correct answer from the options given below:
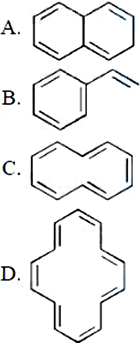
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 40
Which of the following are aromatic?
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 41
Given below are two statements:
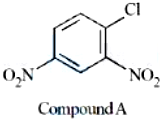
Statement I: IUPAC name of Compound A is 4-chloro-1,3-dinitrobenzene.
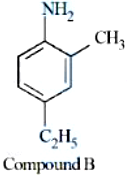
Statement II: IUPAC name of Compound B is 4-ethyl-2-methylaniline. In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 42
In the given compound, the number of $$2°$$ carbon atom/s is _______.
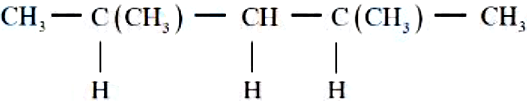
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 43
Iron (III) catalyses the reaction between iodide and persulphate ions, in which
A. $$Fe^{3+}$$ oxidises the iodide ion
B. $$Fe^{3+}$$ oxidises the persulphate ion
C. $$Fe^{2+}$$ reduces the iodide ion
D. $$Fe^{2+}$$ reduces the persulphate ion.
Choose the most appropriate answer from the options given below:
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 44
Number of Complexes with even number of electrons in $$t_{2g}$$ orbitals is - $$[Fe(H_2O)_6]^{2+}$$, $$[Co(H_2O)_6]^{2+}$$, $$[Co(H_2O)_6]^{3+}$$, $$[Cu(H_2O)_6]^{2+}$$, $$[Cr(H_2O)_6]^{2+}$$
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 45
An octahedral complex with the formula $$CoCl_3 \cdot nNH_3$$ upon reaction with excess of $$AgNO_3$$ solution gives 2 moles of $$AgCl$$. Consider the oxidation state of $$Co$$ in the complex is '$$x$$'. The value of "$$x + n$$" is ______
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 46
Which among the following compounds will undergo fastest $$S_N2$$ reaction.
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 47
Identify the major products A and B respectively in the following set of reactions.
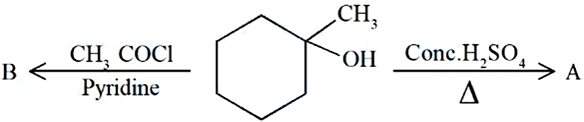
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 48
Identify the product (P) in the following reaction:
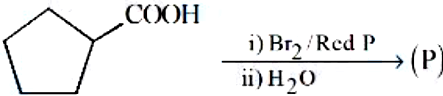
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 49
Match List I with List II
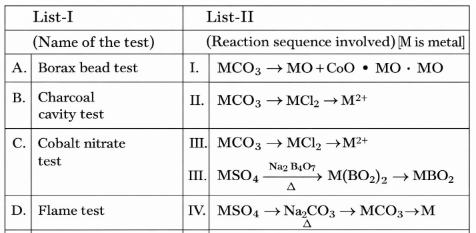
Choose the correct answer from the options given below:
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 50
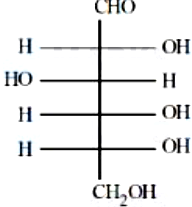
The incorrect statement regarding the given structure is
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 51
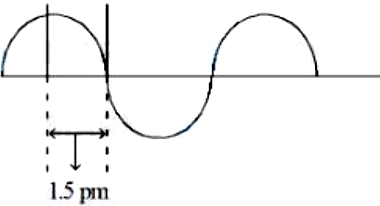
A hypothetical electromagnetic wave is shown below. The frequency of the wave is $$x \times 10^{19}$$ Hz. $$x =$$ ______ (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 52
Number of molecules from the following which are exceptions to octet rule is ______ $$CO_2$$, $$NO_2$$, $$H_2SO_4$$, $$BF_3$$, $$CH_4$$, $$SiF_4$$, $$ClO_2$$, $$PCl_5$$, $$BeF_2$$, $$C_2H_6$$, $$CHCl_3$$, $$CBr_4$$
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 53
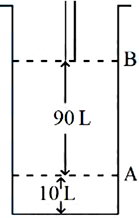
Consider the figure provided. 1 mol of an ideal gas is kept in a cylinder, fitted with a piston, at the position A, at $$18°C$$. If the piston is moved to position B, keeping the temperature unchanged, then '$$x$$' L atm work is done in this reversible process. $$x =$$ ______ L atm. (nearest integer) [Given: Absolute temperature $$= °C + 273.15$$, $$R = 0.08206 \text{ L atm mol}^{-1} \text{K}^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 54
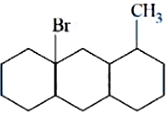
The number of optical isomers in following compound is: ________
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 55
A solution containing $$10 \text{ g}$$ of an electrolyte $$AB_2$$ in $$100 \text{ g}$$ of water boils at $$100.52°C$$. The degree of ionization of the electrolyte $$(\alpha)$$ is ______ $$\times 10^{-1}$$. (nearest integer) [Given: Molar mass of $$AB_2 = 200 \text{ g mol}^{-1}$$, $$K_b$$ (molal boiling point elevation const. of water) $$= 0.52 \text{ K kg mol}^{-1}$$, boiling point of water $$= 100°C$$; $$AB_2$$ ionises as $$AB_2 \rightarrow A^{2+} + 2B^-$$]
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 56
Consider the following reaction $$A + B \rightarrow C$$. The time taken for A to become $$1/4^{th}$$ of its initial concentration is twice the time taken to become $$1/2$$ of the same. Also, when the change of concentration of $$B$$ is plotted against time, the resulting graph gives a straight line with a negative slope and a positive intercept on the concentration axis. The overall order of the reaction is ________
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 57
The 'spin only' magnetic moment value of $$MO_4^{2-}$$ is ______ BM. (Where M is a metal having least metallic radii among $$Sc, Ti, V, Cr, Mn$$ and $$Zn$$). (Given atomic number: $$Sc = 21, Ti = 22, V = 23, Cr = 24, Mn = 25$$ and $$Zn = 30$$)
789
456
123
0.-
Clear All
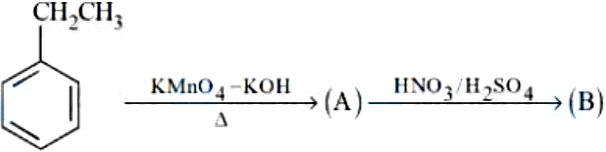
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 58
Major product $$B$$ of the following reaction has ______ $$\pi$$-bond.
789
456
123
0.-
Clear All
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 59
If $$279 \text{ g}$$ of aniline is reacted with one equivalent of benzenediazonium chloride, the maximum amount of aniline yellow formed will be ______ g. (nearest integer) (consider complete conversion).
789
456
123
0.-
Clear All
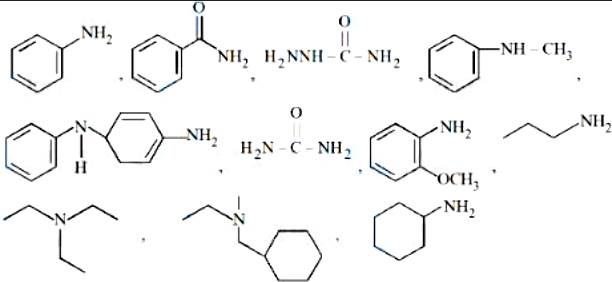
NTA JEE Mains 8th April 2024 Shift 1 - Chemistry - Question 60
Number of amine compounds from the following giving solids which are soluble in NaOH upon reaction with Hinsberg's reagent is ________
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




