Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Among the following halogens $$F_2, Cl_2, Br_2$$ and $$I_2$$. Which can undergo disproportionation reactions?
Which halogens can undergo disproportionation reactions?
Disproportionation is a reaction where the same element is simultaneously oxidized and reduced. For halogens in their elemental form (oxidation state 0), disproportionation requires the element to form both positive and negative oxidation states.
The analysis of each halogen is as follows.
$$F_2$$: Fluorine is the most electronegative element and can only exist in oxidation states 0 and $$-1$$. It cannot achieve positive oxidation states, so it cannot disproportionate.
$$Cl_2$$: Can form $$-1$$ (in $$Cl^-$$) and positive states like $$+1$$ (in $$OCl^-$$). For example: $$Cl_2 + 2NaOH \rightarrow NaCl + NaOCl + H_2O$$. It can disproportionate.
$$Br_2$$: Similarly, can form $$-1$$ and positive oxidation states. It can disproportionate.
$$I_2$$: Can form $$-1$$ and positive states (e.g., $$+5$$ in $$IO_3^-$$). It can disproportionate.
The correct answer is Option (4): $$Cl_2$$, $$Br_2$$ and $$I_2$$.
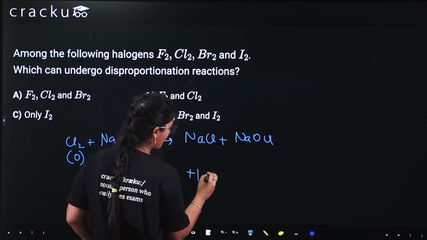
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
