Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
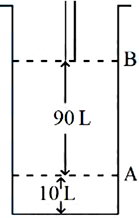
Consider the figure provided. 1 mol of an ideal gas is kept in a cylinder, fitted with a piston, at the position A, at $$18°C$$. If the piston is moved to position B, keeping the temperature unchanged, then '$$x$$' L atm work is done in this reversible process. $$x =$$ ______ L atm. (nearest integer) [Given: Absolute temperature $$= °C + 273.15$$, $$R = 0.08206 \text{ L atm mol}^{-1} \text{K}^{-1}$$]
Correct Answer: 55
For an isothermal reversible expansion, the magnitude of work done ($$W$$) is given by:
$$W = 2.303 \, nRT \log_{10}\left(\frac{V_2}{V_1}\right)$$
Substitute the given values into the formula:
$$x = 2.303 \times 1 \times 0.08206 \times 291.15 \times \log_{10}\left(\frac{100}{10}\right)$$
Since $$\log_{10}(10) = 1$$:
$$x = 2.303 \times 0.08206 \times 291.15 \times 1$$
$$x \approx 55.02\text{ L atm}$$
Rounding to the nearest integer, $$x = 55$$.

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
