Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
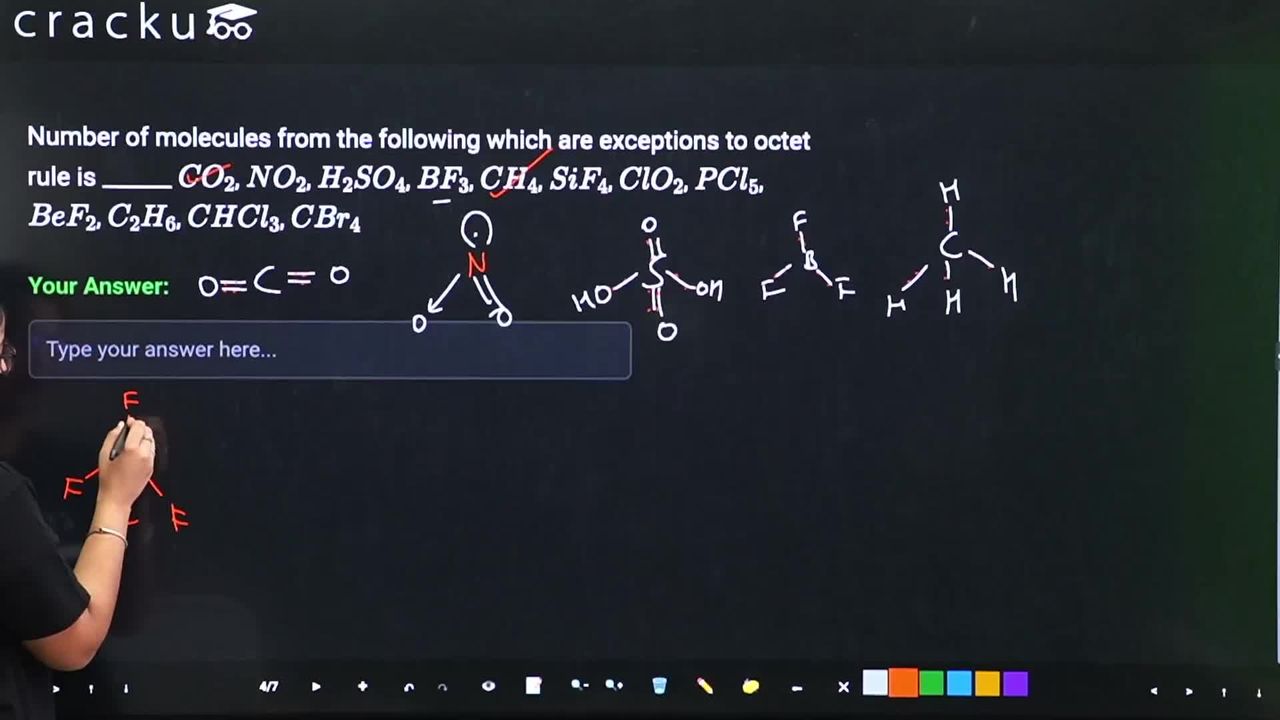
Number of molecules from the following which are exceptions to octet rule is ______ $$CO_2$$, $$NO_2$$, $$H_2SO_4$$, $$BF_3$$, $$CH_4$$, $$SiF_4$$, $$ClO_2$$, $$PCl_5$$, $$BeF_2$$, $$C_2H_6$$, $$CHCl_3$$, $$CBr_4$$
Correct Answer: 6
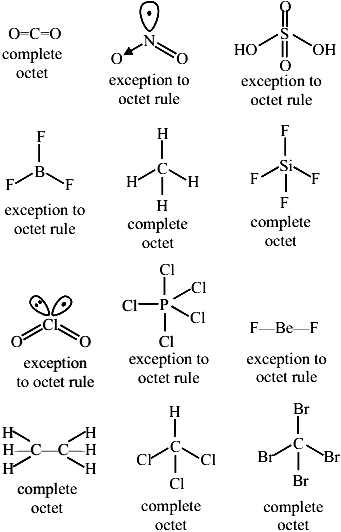
These central atoms follow the standard rule by sharing electrons until they reach a stable count of 8.
These central atoms are stable even though they lack a full shell of 8 electrons.
Elements in Period 3 or below have empty $$d$$-orbitals that allow them to hold more than 8 electrons.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
