Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
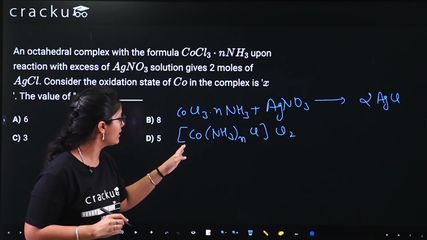
An octahedral complex with the formula $$CoCl_3 \cdot nNH_3$$ upon reaction with excess of $$AgNO_3$$ solution gives 2 moles of $$AgCl$$. Consider the oxidation state of $$Co$$ in the complex is '$$x$$'. The value of "$$x + n$$" is ______
$$CoCl_3 \cdot nNH_3$$ gives 2 mol AgCl with excess AgNO₃. Find $$x + n$$ where $$x$$ is the oxidation state of Co.
Since 2 mol AgCl is produced, 2 Cl⁻ ions are outside the coordination sphere and 1 Cl is inside as a ligand, giving the formula $$[Co(NH_3)_nCl]Cl_2$$.
The charge on the complex ion is +2, where the inner Cl carries a -1 charge and NH₃ is neutral, so $$x + 0 \times n + (-1) = +2 \implies x = +3$$.
Assuming octahedral geometry with coordination number 6 and ligands consisting of n NH₃ molecules plus 1 Cl, we have $$n + 1 = 6$$, hence $$n = 5$$.
Therefore, $$x + n = 3 + 5 = 8$$.
The correct answer is Option (2): 8.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
