NTA JEE Main 8th April 2019 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 31
The quantum number of four electrons are given below:
I. $$n = 4, l = 2, m_l = -2, m_s = -1/2$$
II. $$n = 3, l = 2, m_l = 1, m_s = +1/2$$
III. $$n = 4, l = 1, m_l = 0, m_s = +1/2$$
IV. $$n = 3, l = 1, m_l = 1, m_s = -1/2$$
The correct order of their increasing energies will be:
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 32
The size of the iso-electronic species Cl$$^{-}$$, Ar and Ca$$^{2+}$$ is affected by:
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 33
Which one of the following equations does not correctly represent the first law of thermodynamics for the given processes involving an ideal gas? (Assume non-expansion work is zero)
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 34
For silver, $$C_p$$ (J K$$^{-1}$$ mol$$^{-1}$$) = 23 + 0.01T. If the temperature T of 3 moles of silver is raised from 300 K to 1000 K at 1 atm pressure, the value of $$\Delta H$$ will be close to:
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 35
If solubility product of Zr$$_3$$(PO$$_4$$)$$_4$$ is denoted by K$$_{sp}$$ and its molar solubility is denoted by S, then which of the following relation between S and K$$_{sp}$$ is correct?
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 36
In order to oxidize a mixture of one mole of each of FeC$$_2$$O$$_4$$, Fe$$_2$$(C$$_2$$O$$_4$$)$$_3$$, FeSO$$_4$$ and Fe$$_2$$(SO$$_4$$)$$_3$$ in acidic medium, the number of moles of KMnO$$_4$$ is:
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 37
Given that,
$$E^{0}_{O_2/H_2O} = +1.23$$ V;
$$E^{0}_{S_2O_8^{2-}/SO_4^{2-}} = 2.05$$ V;
$$E^{0}_{Br_2/Br^{-}} = +1.09$$ V;
$$E^{0}_{Au^{3+}/Au} = 1.4$$ V
The strongest oxidizing agent is:
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 38
100 mL of a water sample contains 0.81 g of calcium bicarbonate and 0.73 g of magnesium bicarbonate. The hardness of this water sample expressed in terms of equivalents of CaCO$$_3$$ is: (molar mass of calcium bicarbonate is 162 g mol$$^{-1}$$ and magnesium bicarbonate is 146 g mol$$^{-1}$$)
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 39
The correct order of hydration enthalpies of alkali metal ions is:
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 40
Diborane B$$_2$$H$$_6$$ reacts independently with O$$_2$$ and H$$_2$$O to produce, respectively:
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 41
The IUPAC name of the following compound is:
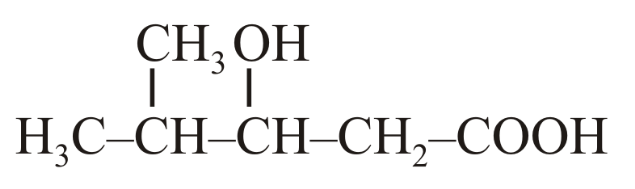
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 42
The major product of the following reaction is:
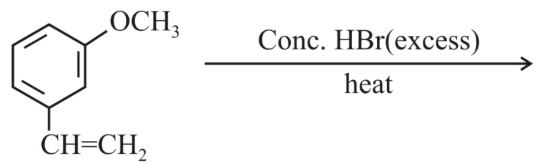
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 43
The major product of the following reaction is:
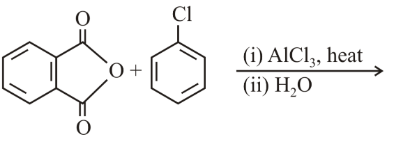
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 44
Which is wrong with respect to our responsibility as a human being to protect our environment?
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 45
Assertion: Ozone is destroyed by CFCs in the upper stratosphere.
Reason: Ozone holes increase the amount of UV radiation reaching the earth.
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 46
Element B forms ccp structure and A occupies half of the octahedral voids, while oxygen atoms occupy all the tetrahedral voids. The structure of bimetallic oxide is:
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 47
The vapour pressures of pure liquids A and B are 400 and 600 mm Hg respectively at 298 K. On mixing the two liquids, the sum of their volumes is equal to the volume of the final mixture. The mole fraction of liquid B is 0.5 in the mixture. The vapour pressure of the final solution, the mole fractions of components A and B in the vapour phase, respectively are:
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 48
For the reaction 2A + B $$\rightarrow$$ C, the values of initial rate at different reactant concentrations are given in the table below.
[A] (mol L$$^{-1}$$) [B] (mol L$$^{-1}$$) Initial Rate (mol L$$^{-1}$$s$$^{-1}$$)
0.05 0.05 0.045
0.10 0.05 0.090
0.20 0.10 0.72
The rate law for the reactions is:
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 49
Adsorption of a gas follows Freundlich adsorption isotherm. x is the mass of the gas adsorbed on mass m of the adsorbent. The plot of $$\log\frac{x}{m}$$ vs $$\log p$$ is shown in the given graph. $$\frac{x}{m}$$ is proportional to:
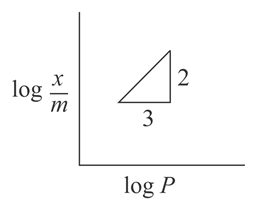
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 50
Which respect to an ore, Ellingham diagram helps to predict the feasibility of its:
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 51
The lanthanide ion that would show colour is:
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 52
The correct order of the spin-only magnetic moment of metal ions in the following low-spin complexes, [V(CN)$$_6$$]$$^{4-}$$, [Fe(CN)$$_6$$]$$^{4-}$$, [Ru(NH$$_3$$)$$_6$$]$$^{3+}$$ and [Cr(NH$$_3$$)$$_6$$]$$^{2+}$$, is:
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 53
The following ligand is:
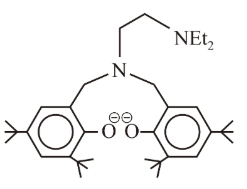
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 54
An organic compound neither reacts with neutral ferric chloride solution nor with Fehling solution. It however, reacts with Grignard reagent and gives positive iodoform test. The compound is:
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 55
The major product of the following reaction is:
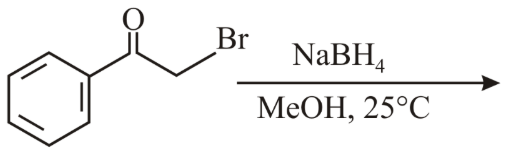
NaBH$$_4$$, MeOH, 25°C
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 56
An organic compound 'X' showing the following solubility profile is:
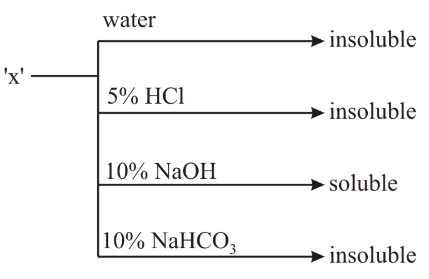
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 57
Which of the following amines can be prepared by Gabriel phthalimide reaction?
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 58
Coupling of benzene diazonium chloride with 1-naphthol in alkaline medium will give:
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 59
In the following compounds, the decreasing order of basic strength will be:
NTA JEE Main 8th April 2019 Shift 1 - Chemistry - Question 60
Maltose on treatment with dilute HCl gives:
.webp)
.webp)
.webp)
.webp)




