NTA JEE Mains 5th April 2024 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 31
An organic compound has $$42.1\%$$ carbon, $$6.4\%$$ hydrogen and remainder is oxygen. If its molecular weight is 342, then its molecular formula is :
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 32
The incorrect postulates of the Dalton's atomic theory are : (A) Atoms of different elements differ in mass. (B) Matter consists of divisible atoms. (C) Compounds are formed when atoms of different element combine in a fixed ratio. (D) All the atoms of given element have different properties including mass. (E) Chemical reactions involve reorganisation of atoms. Choose the correct answer from the options given below :
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 33
Given below are two statements :
Statement I : In group 13, the stability of +1 oxidation state increases down the group.
Statement II : The atomic size of gallium is greater than that of aluminium.
In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 34
The statement(s) that are correct about the species $$O^{2-}, F^-, Na^+$$ and $$Mg^{2+}$$. (A) All are isoelectronic (B) All have the same nuclear charge (C) $$O^{2-}$$ has the largest ionic radii (D) $$Mg^{2+}$$ has the smallest ionic radii. Choose the most appropriate answer from the options given below :
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 35
Given below are two statements : One is labelled as Assertion (A) and the other is labelled as Reason (R). Assertion (A) : Enthalpy of neutralisation of strong monobasic acid with strong monoacidic base is always $$-57 \text{ kJ mol}^{-1}$$. Reason (R) : Enthalpy of neutralisation is the amount of heat liberated when one mole of $$H^+$$ ions furnished by acid combine with one mole of $$OH^-$$ ions furnished by base to form one mole of water. In the light of the above statements, choose the correct answer from the options given below.
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 36
The following reaction occurs in the Blast furnace where iron ore is reduced to iron metal $$Fe_2O_{3(s)} + 3CO_{(g)} \rightleftharpoons Fe_{(l)} + 3CO_{2(g)}$$. Using the Le-Chatelier's principle, predict which one of the following will not disturb the equilibrium.
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 37
The number of neutrons present in the more abundant isotope of boron is '$$x$$'. Amorphous boron upon heating with air forms a product, in which the oxidation state of boron is '$$y$$'. The value of $$x + y$$ is ______
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 38
Number of $$\sigma$$ and $$\pi$$ bonds present in ethylene molecule is respectively :
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 39
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R). Assertion (A) : Cis form of alkene is found to be more polar than the trans form. Reason (R) : Dipole moment of trans isomer of 2-butene is zero. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 40
For the Compounds : (A) $$H_3C-CH_2-O-CH_2-CH_2-CH_3$$
(B) $$H_3C-CH_2-CH_2-CH_2-CH_2-CH_3$$ (
C)
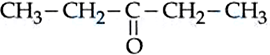
(D)
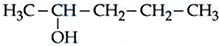
The increasing order of boiling point is :
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 41
Given below are two statements: Statement I : Nitration of benzene involves the following step
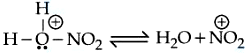
Statement II : Use of Lewis base promotes the electrophilic substitution of benzene. In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 42
The reaction at cathode in the cells commonly used in clocks involves.
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 43
Molar ionic conductivities of divalent cation and anion are $$57 \text{ S cm}^2 \text{ mol}^{-1}$$ and $$73 \text{ S cm}^2 \text{ mol}^{-1}$$ respectively. The molar conductivity of solution of an electrolyte with the above cation and anion will be :
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 44
The metal that shows highest and maximum number of oxidation state is :
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 45
Which one of the following complexes will exhibit the least paramagnetic behaviour? [Atomic number, $$Cr = 24, Mn = 25, Fe = 26, Co = 27$$]
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 46
The correct order of ligands arranged in increasing field strength.
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 47
Given below are two statements: Statement I : Bromination of phenol in solvent with low polarity such as $$CHCl_3$$ or $$CS_2$$ requires Lewis acid catalyst. Statement II : The Lewis acid catalyst polarises the bromine to generate $$Br^+$$. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 48
Identify compound (Z) in the following reaction sequence.
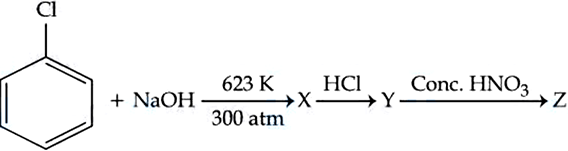
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 49
Identify '$$A$$' in the following reaction:
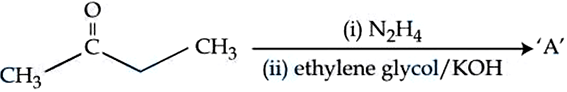
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 50
Which of the following gives a positive test with ninhydrin?
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 51
$$9.3 \text{ g}$$ of pure aniline is treated with bromine water at room temperature to give a white precipitate of the product '$$P$$'. The mass of product '$$P$$' obtained is $$26.4 \text{ g}$$. The percentage yield is ______ %.
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 52
The value of Rydberg constant $$(R_H)$$ is $$2.18 \times 10^{-18} \text{ J}$$. The velocity of electron having mass $$9.1 \times 10^{-31} \text{ kg}$$ in Bohr's first orbit of hydrogen atom $$=$$ ______ $$\times 10^5 \text{ ms}^{-1}$$ (nearest integer).
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 53
In the Lewis dot structure for $$NO_2^-$$, total number of valence electrons around nitrogen is ______
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 54
The heat of combustion of solid benzoic acid at constant volume is $$-321.30 \text{ kJ}$$ at $$27°C$$. The heat of combustion at constant pressure is $$(-321.30 - xR) \text{ kJ}$$, the value of $$x$$ is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 55
An artificial cell is made by encapsulating $$0.2 \text{ M}$$ glucose solution within a semipermeable membrane. The osmotic pressure developed when the artificial cell is placed within a $$0.05 \text{ M NaCl}$$ solution at $$300 \text{ K}$$ is ______ $$\times 10^{-1} \text{ bar}$$. (nearest integer). [Given : $$R = 0.083 \text{ L bar mol}^{-1} \text{ K}^{-1}$$] Assume complete dissociation of $$NaCl$$
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 56
During Kinetic study of reaction $$2A + B \to C + D$$, the following results were obtained :
I: A = 0.1 M, B = 0.1 M, initial rate of formation of D = $$6.0 \times 10^{-3}$$
II: A = 0.3 M, B = 0.2 M, initial rate = $$7.20 \times 10^{-2}$$
III: A = 0.3 M, B = 0.4 M, initial rate = $$2.88 \times 10^{-1}$$
IV: A = 0.4 M, B = 0.1 M, initial rate = $$2.40 \times 10^{-2}$$
Based on above data, overall order of the reaction is ______
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 57
The spin-only magnetic moment value of the ion among $$Ti^{2+}, V^{2+}, Co^{3+}$$ and $$Cr^{2+}$$, that acts as strong oxidising agent in aqueous solution is ______ BM (Near integer). (Given atomic numbers : $$Ti : 22, V : 23, Cr : 24, Co : 27$$)
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 58
The number of halobenzenes from the following that can be prepared by Sandmeyer's reaction is ______
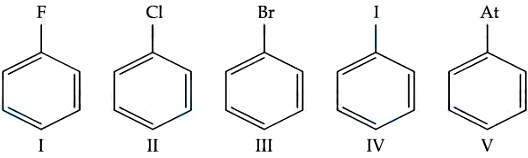
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 59
Consider the given chemical reaction sequence :
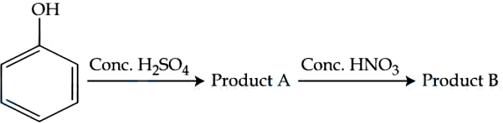
Total sum of oxygen atoms in Product A and Product B are ______
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Chemistry - Question 60
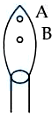
In a borax bead test under hot condition, a metal salt (one from the given) is heated at point B of the flame, resulted in green colour salt bead. The spin-only magnetic moment value of the salt is ______ BM (Nearest integer) [Given atomic number of $$Cu = 29, Ni = 28, Mn = 25, Fe = 26$$]
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




