Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
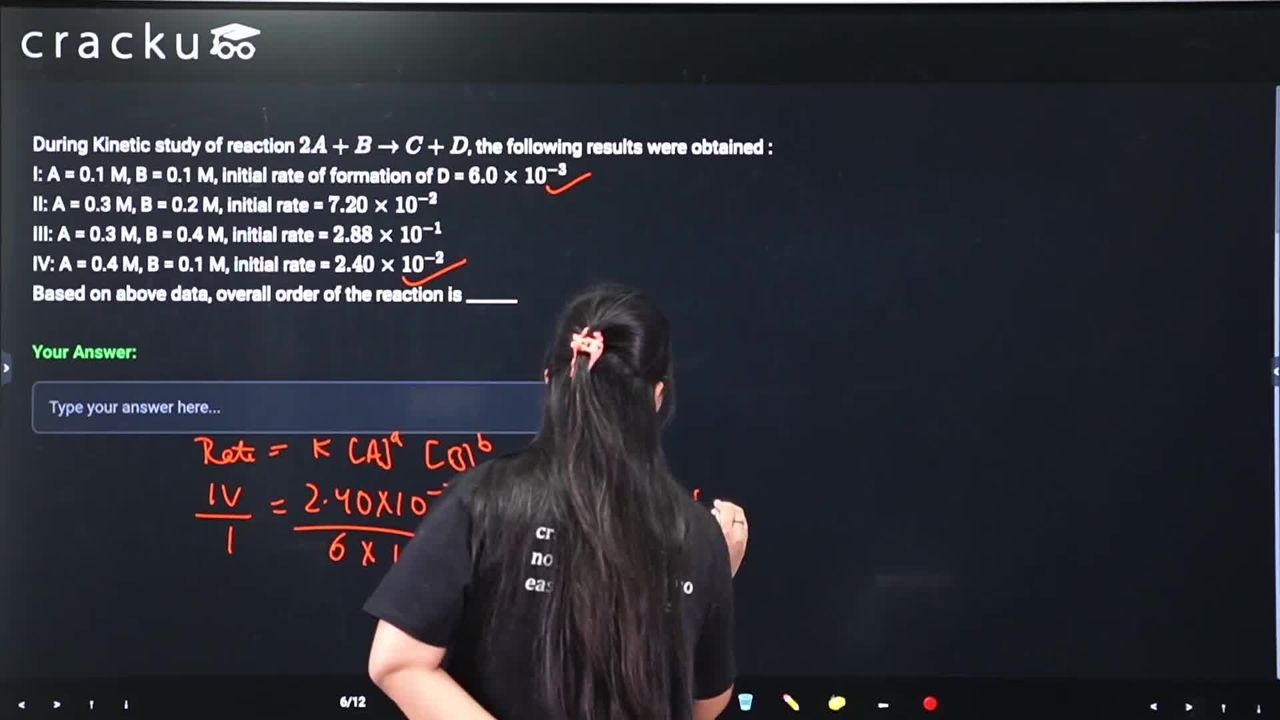
During Kinetic study of reaction $$2A + B \to C + D$$, the following results were obtained :
I: A = 0.1 M, B = 0.1 M, initial rate of formation of D = $$6.0 \times 10^{-3}$$
II: A = 0.3 M, B = 0.2 M, initial rate = $$7.20 \times 10^{-2}$$
III: A = 0.3 M, B = 0.4 M, initial rate = $$2.88 \times 10^{-1}$$
IV: A = 0.4 M, B = 0.1 M, initial rate = $$2.40 \times 10^{-2}$$
Based on above data, overall order of the reaction is ______
Correct Answer: 3
For the reaction $$2A + B \rightarrow C + D$$, let $$\text{Rate} = k[A]^a[B]^b$$.
From experiments I and IV (where B is constant at 0.1 M):
$$ \frac{r_{IV}}{r_I} = \left(\frac{0.4}{0.1}\right)^a = 4^a = \frac{2.40 \times 10^{-2}}{6.0 \times 10^{-3}} = 4 $$
$$ 4^a = 4 \Rightarrow a = 1 $$
From experiments II and III (where A is constant at 0.3 M):
$$ \frac{r_{III}}{r_{II}} = \left(\frac{0.4}{0.2}\right)^b = 2^b = \frac{2.88 \times 10^{-1}}{7.20 \times 10^{-2}} = 4 $$
$$ 2^b = 4 \Rightarrow b = 2 $$
Thus, the overall order of the reaction is $$a + b = 1 + 2 = 3$$.
Answer: 3.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
