Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
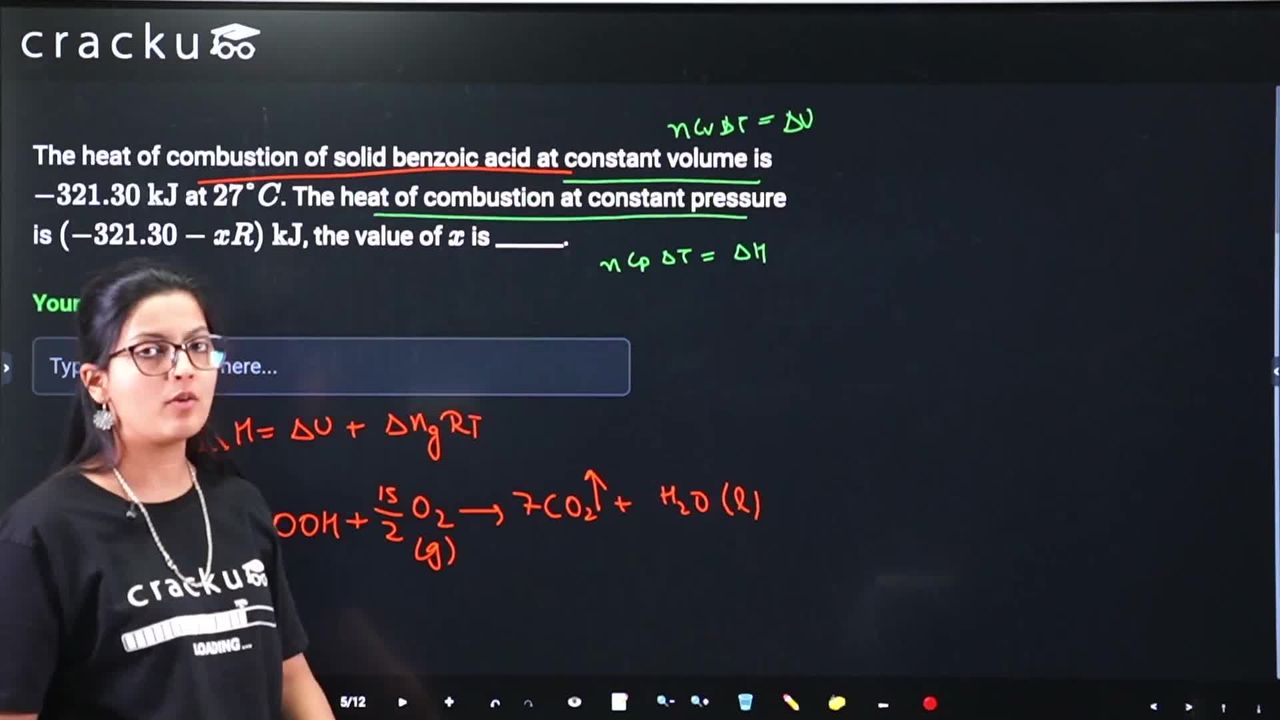
The heat of combustion of solid benzoic acid at constant volume is $$-321.30 \text{ kJ}$$ at $$27°C$$. The heat of combustion at constant pressure is $$(-321.30 - xR) \text{ kJ}$$, the value of $$x$$ is ______.
Correct Answer: 150
The combustion of benzoic acid is represented by the reaction $$C_6H_5COOH(s) + \frac{15}{2}O_2(g) \rightarrow 7CO_2(g) + 3H_2O(l)$$.
The relation between heat at constant pressure and constant volume is given by $$\Delta H = \Delta U + \Delta n_g RT$$, where $$\Delta n_g$$ is the difference between the moles of gaseous products and reactants. Calculating, $$\Delta n_g = 7 - \frac{15}{2} = -\frac{1}{2}$$.
Substituting this value into the relation yields $$\Delta H = \Delta U + \left(-\frac{1}{2}\right)RT = -321.30 - \frac{1}{2}RT$$.
At $$T = 27°C = 300$$ K, the enthalpy change becomes $$\Delta H = -321.30 - \frac{1}{2}(R)(300) = -321.30 - 150R$$, from which it follows that $$x = 150$$.
Therefore, the value of x is 150.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
