Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
We need to find the correct order of ligands arranged in increasing field strength according to the spectrochemical series.
The spectrochemical series (increasing field strength) is:
$$I^- < Br^- < S^{2-} < Cl^- < N_3^- < F^- < OH^- < ox^{2-} < H_2O < NCS^- < CH_3CN < py < NH_3 < en < bipy < phen < NO_2^- < PPh_3 < CN^- < CO < NO^+$$
Check each option:
Option A: $$F^- < Br^- < I^- < NH_3$$ - Incorrect. The order among halides is $$I^- < Br^- < Cl^- < F^-$$, not $$F^- < Br^- < I^-$$.
Option B: $$Br^- < F^- < H_2O < NH_3$$ - This follows the spectrochemical series correctly: $$Br^-$$ is weaker than $$F^-$$, $$F^-$$ is weaker than $$H_2O$$, and $$H_2O$$ is weaker than $$NH_3$$. This is correct.
Option C: $$H_2O < OH^- < CN^- < NH_3$$ - Incorrect. $$CN^-$$ is a stronger field ligand than $$NH_3$$, so $$NH_3 < CN^-$$, not $$CN^- < NH_3$$.
Option D: $$Cl^- < OH^- < Br^- < CN^-$$ - Incorrect. $$Br^-$$ is a weaker field ligand than $$Cl^-$$, so $$Br^-$$ should come before $$Cl^-$$.
The correct answer is Option B.
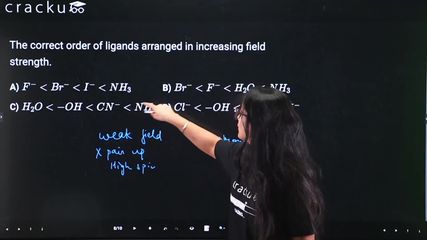
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
