Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The statement(s) that are correct about the species $$O^{2-}, F^-, Na^+$$ and $$Mg^{2+}$$. (A) All are isoelectronic (B) All have the same nuclear charge (C) $$O^{2-}$$ has the largest ionic radii (D) $$Mg^{2+}$$ has the smallest ionic radii. Choose the most appropriate answer from the options given below :
Species: $$O^{2-}, F^-, Na^+, Mg^{2+}$$.
(A) All are isoelectronic: All have 10 electrons. $$O^{2-}$$ (8+2=10), $$F^-$$ (9+1=10), $$Na^+$$ (11-1=10), $$Mg^{2+}$$ (12-2=10). Correct. ✓
(B) All have the same nuclear charge: $$O$$ has 8 protons, $$F$$ has 9, $$Na$$ has 11, $$Mg$$ has 12. They have different nuclear charges. Incorrect. ✗
(C) $$O^{2-}$$ has the largest ionic radius: For isoelectronic species, the one with the smallest nuclear charge has the largest radius. $$O^{2-}$$ has the smallest nuclear charge (8), so it has the largest ionic radius. Correct. ✓
(D) $$Mg^{2+}$$ has the smallest ionic radius: $$Mg^{2+}$$ has the largest nuclear charge (12) among the isoelectronic species, so it has the smallest ionic radius. Correct. ✓
Correct statements: (A), (C), and (D).
The correct answer is Option (3): (A), (C) and (D) only.
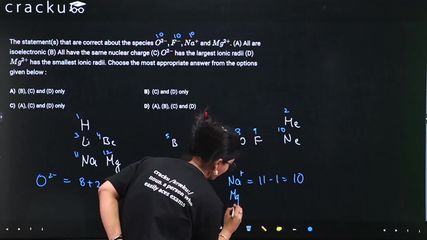
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
