NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 31
The correct sequence of electron gain enthalpy of the elements listed below is A. Ar B. Br C. F D. S. Choose the most appropriate from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 32
The linear combination of atomic orbitals to form molecular orbitals takes place only when the combining atomic orbitals A. have the same energy B. have the minimum overlap C. have same symmetry about the molecular axis D. have different symmetry about the molecular axis. Choose the most appropriate from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 33
For the given reaction, choose the correct expression of $$K_C$$ from the following: $$\text{Fe}^{3+}_{(aq)} + \text{SCN}^-_{(aq)} \rightleftharpoons (\text{FeSCN})^{2+}_{(aq)}$$
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 34
Consider the oxides of group 14 elements $$SiO_2, GeO_2, SnO_2, PbO_2, CO$$ and $$GeO$$. The amphoteric oxides are
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 35
A species having carbon with sextet of electrons and can act as electrophile is called
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 36
'Adsorption' principle is used for which of the following purification method?
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 37
Given below are two statements: Statement I: IUPAC name of $$HO - CH_2 - (CH_2)_3 - CH_2 - COCH_3$$ is 7-hydroxyheptan-2-one. Statement II: 2-oxoheptan-7-ol is the correct IUPAC name for the above compound. In the light of the above statements, Choose the most appropriate answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 38
Match List I with List II:

Choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 39
The product (C) in the below mentioned reaction is: $$CH_3-CH_2-CH_2-Br \xrightarrow[\Delta]{KOH_{alc}} A \xrightarrow{HBr} B \xrightarrow[\Delta]{KOH_{aq}} C$$
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 40
Identify the mixture that shows positive deviations from Raoult's Law
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 41
The metals that are employed in the battery industries are A. Fe, B. Mn, C. Ni, D. Cr, E. Cd. Choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 42
Identify the factor from the following that does not affect electrolytic conductance of a solution.
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 43
Integrated rate law equation for a first order gas phase reaction is given by (where $$P_i$$ is initial pressure and $$P_t$$ is total pressure at time $$t$$)
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 44
Give below are two statements:
Statement-I: Noble gases have very high boiling points.
Statement-II: Noble gases are monoatomic gases. They are held together by strong dispersion forces. Because of this they are liquefied at very low temperature. Hence, they have very high boiling points.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 45
Identify correct statements from below:
A. The chromate ion is square planar.
B. Dichromates are generally prepared from chromates.
C. The green manganate ion is diamagnetic.
D. Dark green coloured $$K_2MnO_4$$ disproportionates in a neutral or acidic medium to give permanganate.
E. With increasing oxidation number of transition metal, ionic character of the oxides decreases.
Choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 46
The correct statements from the following are:
A. The strength of anionic ligands can be explained by crystal field theory.
B. Valence bond theory does not give a quantitative interpretation of kinetic stability of coordination compounds.
C. The hybridization involved in formation of $$[Ni(CN)_4]^{2-}$$ complex is $$dsp^2$$.
D. The number of possible isomer(s) of cis-$$[PtCl_2(en)_2]^{2+}$$ is one.
Choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 47
Given below are two statements:
Assertion A: $$pK_a$$ value of phenol is $$10.0$$ while that of ethanol is $$15.9$$.
Reason R: Ethanol is stronger acid than phenol.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 48
Given below are two statements:
Assertion A: Alcohols react both as nucleophiles and electrophiles.
Reason R: Alcohols react with active metals such as sodium, potassium and aluminum to yield corresponding alkoxides and liberate hydrogen.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 49
The compound that is white in color is
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 50
Match List I with List II:

Choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 51
Number of moles of methane required to produce $$22$$ g $$CO_{2(g)}$$ after combustion is $$x \times 10^{-2}$$ moles. The value of $$x$$ is
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 52
The ionization energy of sodium in $$kJ \text{ mol}^{-1}$$, if electromagnetic radiation of wavelength $$242$$ nm is just sufficient to ionize sodium atom is ______. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 53
The number of species from the following in which the central atom uses $$sp^3$$ hybrid orbitals in its bonding is _________. $$NH_3, SO_2, SiO_2, BeCl_2, CO_2, H_2O, CH_4, BF_3$$
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 54
Consider the following reaction at 298 K. $$\frac{3}{2}O_{2(g)} \rightleftharpoons O_{3(g)}$$, $$K_p = 2.47 \times 10^{-29}$$. $$\Delta_r G^0$$ for the reaction is _________ kJ. (Given $$R = 8.314 \text{ JK}^{-1}\text{mol}^{-1}$$) Round off your answer to the nearest integer.
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 55
Number of alkanes obtained on electrolysis of a mixture of $$CH_3COONa$$ and $$C_2H_5COONa$$ is _____.
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 56
One Faraday of electricity liberates $$x \times 10^{-1}$$ gram atom of copper from copper sulphate, $$x$$ is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 57
The 'Spin only' Magnetic moment for $$[Ni(NH_3)_6]^{2+}$$ is ______ $$\times 10^{-1}$$ BM. (given = Atomic number of Ni : 28) Round off your answer to the nearest integer.
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 58
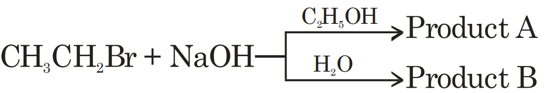
The total number of hydrogen atoms in product A and product B is __________.
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 59
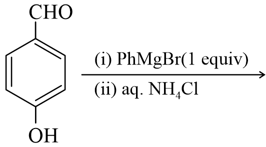
The number of hydroxyl groups present in the product $$P$$ is __________.
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Chemistry - Question 60
Molar mass of the salt from $$NaBr, NaNO_3, KI$$ and $$CaF_2$$ which does not evolve coloured vapours on heating with concentrated $$H_2SO_4$$ is ____ g mol$$^{-1}$$. (Molar mass in g mol$$^{-1}$$: Na: 23, N: 14, K: 39, O: 16, Br: 80, I: 127, F: 19, Ca: 40)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




