Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
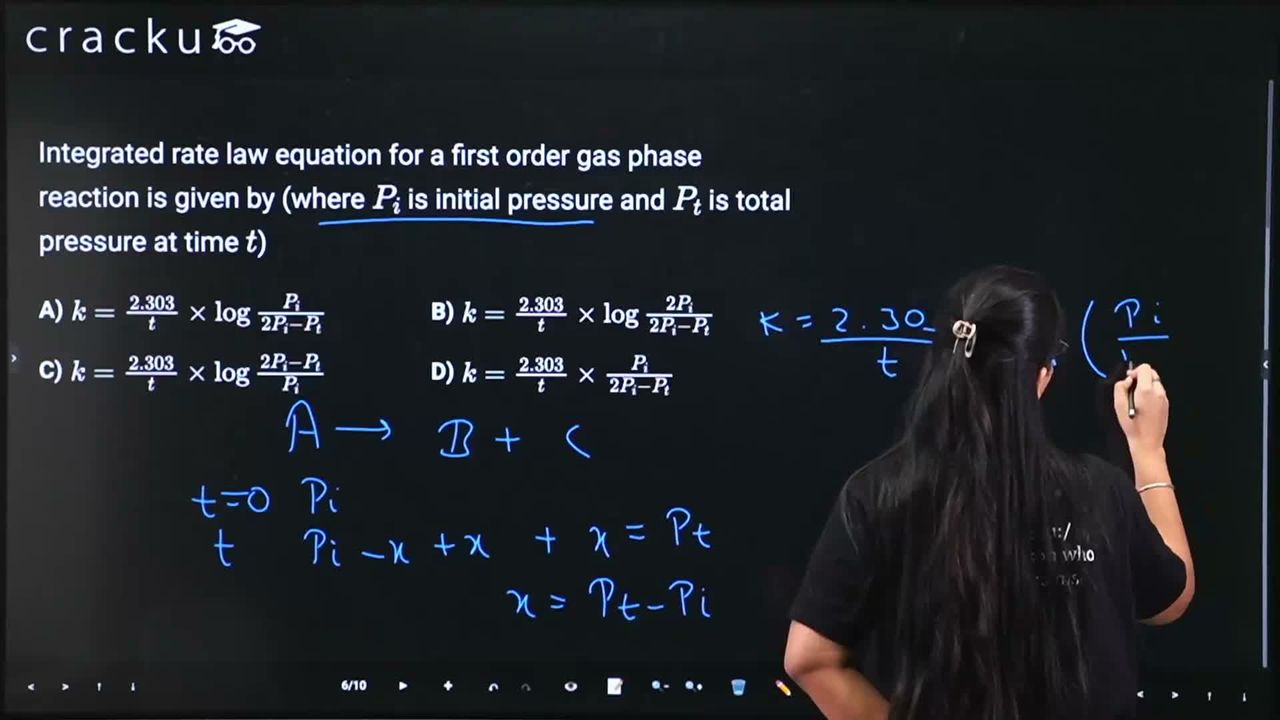
Integrated rate law equation for a first order gas phase reaction is given by (where $$P_i$$ is initial pressure and $$P_t$$ is total pressure at time $$t$$)
For the first-order gas phase reaction $$A(g) \rightarrow B(g) + C(g)$$, we need to find the rate constant expression in terms of pressures.
Set up pressure relationships.
At $$t = 0$$: only A is present with pressure $$P_i$$.
At time $$t$$: let $$x$$ be the decrease in pressure of A. Then:
- Pressure of A: $$P_A = P_i - x$$
- Pressure of B: $$P_B = x$$
- Pressure of C: $$P_C = x$$
- Total pressure: $$P_t = (P_i - x) + x + x = P_i + x$$
Express $$P_A$$ in terms of measurable quantities.
From $$P_t = P_i + x$$: $$x = P_t - P_i$$
$$ P_A = P_i - x = P_i - (P_t - P_i) = 2P_i - P_t $$
Write the first-order rate constant expression.
For a first-order reaction: $$k = \frac{2.303}{t}\log\frac{[A]_0}{[A]_t}$$
In terms of pressures (since pressure is proportional to concentration for ideal gases):
$$ k = \frac{2.303}{t}\log\frac{P_i}{P_A} = \frac{2.303}{t}\log\frac{P_i}{2P_i - P_t} $$
The correct answer is Option (1): $$k = \frac{2.303}{t}\log\frac{P_i}{2P_i - P_t}$$.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
