Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
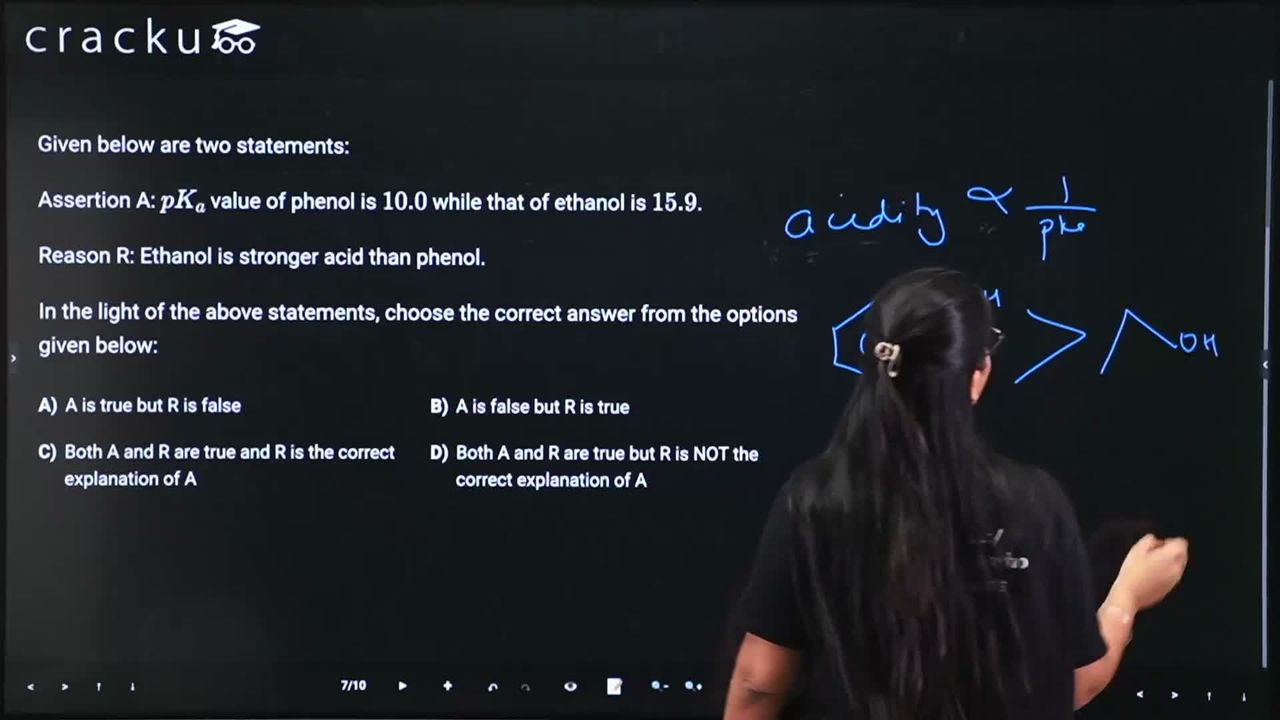
Given below are two statements:
Assertion A: $$pK_a$$ value of phenol is $$10.0$$ while that of ethanol is $$15.9$$.
Reason R: Ethanol is stronger acid than phenol.
In the light of the above statements, choose the correct answer from the options given below:
We need to evaluate the Assertion (A) and Reason (R) about the acidity of phenol and ethanol.
Assertion A: "$$pK_a$$ value of phenol is 10.0 while that of ethanol is 15.9."
This is a factual statement and is TRUE. A lower $$pK_a$$ value indicates a stronger acid. Phenol ($$pK_a = 10.0$$) is a stronger acid than ethanol ($$pK_a = 15.9$$) because the phenoxide ion ($$C_6H_5O^-$$) is stabilized by resonance with the aromatic ring, while the ethoxide ion ($$C_2H_5O^-$$) has no such stabilization.
Reason R: "Ethanol is stronger acid than phenol."
This is FALSE. As explained above, phenol is a much stronger acid than ethanol (lower $$pK_a$$). The statement has the comparison reversed.
The correct answer is Option (1): A is true but R is false.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
