NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 31
Given below are two statements:
Statement - I: Along the period, the chemical reactivity of the element gradually increases from group 1 to group 18.
Statement - II: The nature of oxides formed by group 1 element is basic while that of group 17 elements is acidic.
In the light of the above statements, choose the most appropriate from the options given below:
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 32
Given below are two statements:
Statement-I: Since fluorine is more electronegative than nitrogen, the net dipole moment of $$NF_3$$ is greater than $$NH_3$$.
Statement-II: In $$NH_3$$, the orbital dipole due to lone pair and the dipole moment of NH bonds are in opposite direction, but in $$NF_3$$ the orbital dipole due to lone pair and dipole moments of N-F bonds are in same direction.
In the light of the above statements, choose the most appropriate from the options given below.
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 33
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: $$H_2Te$$ is more acidic than $$H_2S$$.
Reason R: Bond dissociation enthalpy of $$H_2Te$$ is lower than $$H_2S$$.
In the light of the above statements, choose the most appropriate from the options given below.
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 34
IUPAC name of the following compound is:
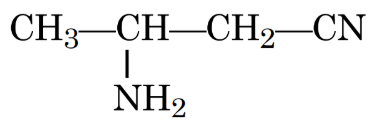
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 35
Which among the following purification methods is based on the principle of "Solubility" in two different solvents?
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 36
The correct stability order of carbocations is
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 37
Product A and B formed in the following set of reactions are:
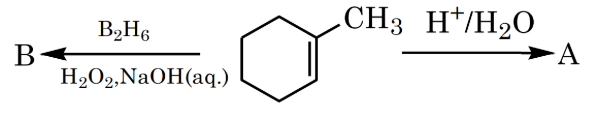
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 38
If a substance '$$A$$' dissolves in solution of a mixture of '$$B$$' and '$$C$$' with their respective number of moles as $$n_A$$, $$n_B$$ and $$n_C$$, mole fraction of $$C$$ in the solution is:
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 39
The solution from the following with highest depression in freezing point/lowest freezing point is
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 40
Reduction potential of ions are given below:
$$ClO_4^-$$: $$E° = 1.19$$ V; $$IO_4^-$$: $$E° = 1.65$$ V; $$BrO_4^-$$: $$E° = 1.74$$ V
The correct order of their oxidising power is:
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 41
Choose the correct statements about the hydrides of group 15 elements.
A. The stability of the hydrides decreases in the order $$NH_3 > PH_3 > AsH_3 > SbH_3 > BiH_3$$
B. The reducing ability of the hydrides increases in the order $$NH_3 < PH_3 < AsH_3 < SbH_3 < BiH_3$$
C. Among the hydrides, $$NH_3$$ is strong reducing agent while $$BiH_3$$ is mild reducing agent.
D. The basicity of the hydrides increases in the order $$NH_3 < PH_3 < AsH_3 < SbH_3 < BiH_3$$
Choose the most appropriate from the options given below:
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 42
The orange colour of $$K_2Cr_2O_7$$ and purple colour of $$KMnO_4$$ is due to
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 43
$$A$$ and $$B$$ formed in the following reactions are:
$$CrO_2Cl_2 + 4NaOH \rightarrow A + 2NaCl + 2H_2O$$
$$A + 2HCl + 2H_2O_2 \rightarrow B + 3H_2O$$
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 44
Alkaline oxidative fusion of $$MnO_2$$ gives "A" which on electrolytic oxidation in alkaline solution produces $$B$$. $$A$$ and $$B$$ respectively are:
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 45
The molecule/ion with square pyramidal shape is:
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 46
The coordination geometry around the manganese in decacarbonyldimanganese(0) is:
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 47
Given below are two statements:
Statement - I: High concentration of strong nucleophilic reagent with secondary alkyl halides which do not have bulky substituents will follow $$S_N2$$ mechanism.
Statement - II: A secondary alkyl halide when treated with a large excess of ethanol follows $$S_N1$$ mechanism.
In the light of the above statements, choose the most appropriate from the options given below:
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 48
Salicylaldehyde is synthesized from phenol, when reacted with
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 49
m-chlorobenzaldehyde on treatment with 50% KOH solution yields
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 50
The products A and B formed in the following reaction scheme are respectively:
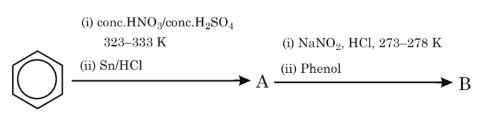
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 51
Number of spectral lines obtained in $$He^+$$ spectra, when an electron makes transition from fifth excited state to first excited state will be
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 52
Two reactions are given below:
$$2Fe_{(s)} + \frac{3}{2}O_{2(g)} \rightarrow Fe_2O_{3(s)}$$, $$\Delta H° = -822$$ kJ/mol
$$C_{(s)} + \frac{1}{2}O_{2(g)} \rightarrow CO_{(g)}$$, $$\Delta H° = -110$$ kJ/mol
Then enthalpy change for following reaction:
$$3C_{(s)} + Fe_2O_{3(s)} \rightarrow 2Fe_{(s)} + 3CO_{(g)}$$
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 53
The pH of an aqueous solution containing 1M benzoic acid ($$pK_a = 4.20$$) and 1M sodium benzoate is 4.5. The volume of benzoic acid solution in 300 mL of this buffer solution is _________ mL.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 54
Total number of species from the following which can undergo disproportionation reaction: $$H_2O_2$$, $$ClO_3^-$$, $$P_4$$, $$Cl_2$$, $$Ag$$, $$Cu^{+1}$$, $$F_2$$, $$NO_2$$, $$K^+$$
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 55
Number of geometrical isomers possible for the given structure is/are ________.
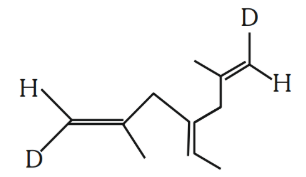
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 56
$$NO_2$$ required for a reaction is produced by decomposition of $$N_2O_5$$ in $$CCl_4$$ as by equation:
$$2N_2O_{5(g)} \rightarrow 4NO_{2(g)} + O_{2(g)}$$
The initial concentration of $$N_2O_5$$ is 3 mol L$$^{-1}$$ and it is 2.75 mol L$$^{-1}$$ after 30 minutes. The rate of formation of $$NO_2$$ is $$x \times 10^{-3}$$ mol L$$^{-1}$$ min$$^{-1}$$, value of $$x$$ is ________.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 57
Number of complexes which show optical isomerism among the following is _________.
$$cis-[Cr(ox)_2Cl_2]^{3-}$$, $$[Co(en)_3]^{3+}$$, $$cis-[Pt(en)_2Cl_2]^{2+}$$, $$cis-[Co(en)_2Cl_2]^+$$, $$trans-[Pt(en)_2Cl_2]^{2+}$$, $$trans-[Cr(ox)_2Cl_2]^{3-}$$
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 58
2-chlorobutane + $$Cl_2 \rightarrow C_4H_8Cl_2$$ (isomers)
Total number of optically active isomers shown by $$C_4H_8Cl_2$$, obtained in the above reaction is ________.
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 59
Number of metal ions characterized by flame test among the following is _________.
$$Sr^{2+}$$, $$Ba^{2+}$$, $$Ca^{2+}$$, $$Cu^{2+}$$, $$Zn^{2+}$$, $$Co^{2+}$$, $$Fe^{2+}$$
789
456
123
0.-
Clear All
NTA JEE Mains 30th Jan 2024 Shift 2 - Chemistry - Question 60
The total number of correct statements, regarding the nucleic acids is _________.
A. RNA is regarded as the reserve of genetic information.
B. DNA molecule self-duplicates during cell division.
C. DNA synthesizes proteins in the cell.
D. The message for the synthesis of particular proteins is present in DNA.
E. Identical DNA strands are transferred to daughter cells.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




