Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
$$NO_2$$ required for a reaction is produced by decomposition of $$N_2O_5$$ in $$CCl_4$$ as by equation:
$$2N_2O_{5(g)} \rightarrow 4NO_{2(g)} + O_{2(g)}$$
The initial concentration of $$N_2O_5$$ is 3 mol L$$^{-1}$$ and it is 2.75 mol L$$^{-1}$$ after 30 minutes. The rate of formation of $$NO_2$$ is $$x \times 10^{-3}$$ mol L$$^{-1}$$ min$$^{-1}$$, value of $$x$$ is ________.
Correct Answer: 17
$$2N_2O_5 \rightarrow 4NO_2 + O_2$$
Rate of disappearance of N$$_2$$O$$_5$$: $$-\frac{\Delta[N_2O_5]}{\Delta t} = \frac{3 - 2.75}{30} = \frac{0.25}{30}$$ mol L$$^{-1}$$ min$$^{-1}$$.
Rate of formation of NO$$_2$$ = $$\frac{4}{2} \times$$ rate of disappearance of N$$_2$$O$$_5$$:
$$= 2 \times \frac{0.25}{30} = \frac{0.5}{30} = \frac{1}{60} \approx 0.01667$$ mol L$$^{-1}$$ min$$^{-1}$$.
$$= 16.67 \times 10^{-3} \approx 17 \times 10^{-3}$$ mol L$$^{-1}$$ min$$^{-1}$$.
Therefore, $$x = \boxed{17}$$.
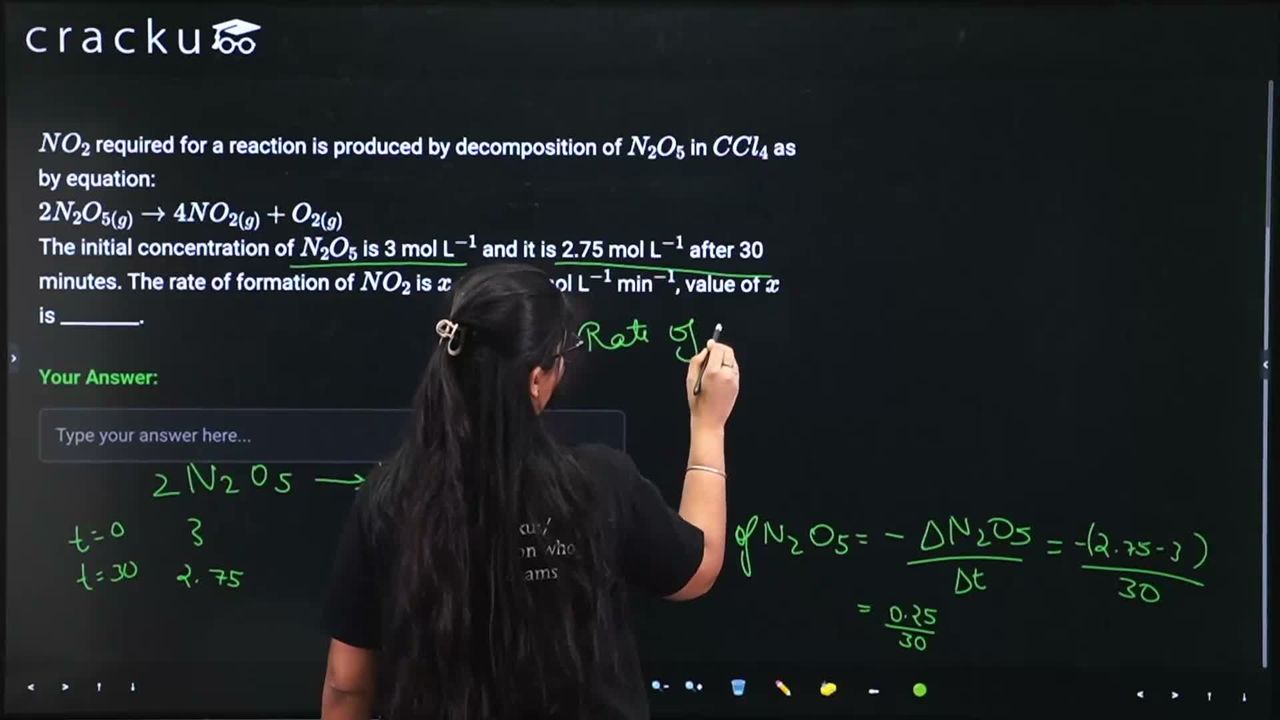
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
