Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
$$A$$ and $$B$$ formed in the following reactions are:
$$CrO_2Cl_2 + 4NaOH \rightarrow A + 2NaCl + 2H_2O$$
$$A + 2HCl + 2H_2O_2 \rightarrow B + 3H_2O$$
Chromyl chloride $$CrO_2Cl_2$$ contains chromium in the $$+6$$ oxidation state. In alkaline medium chloride ions are replaced by oxide ions, converting the molecule into the chromate ion $$CrO_4^{2-}$$.
Writing the reaction with hydroxide ions first:
$$CrO_2Cl_2 + 4OH^- \rightarrow CrO_4^{2-} + 2Cl^- + 2H_2O$$
On adding sodium ions to both sides, the molecular equation becomes
$$CrO_2Cl_2 + 4NaOH \rightarrow Na_2CrO_4 + 2NaCl + 2H_2O$$
Thus, $$A = Na_2CrO_4$$ (sodium chromate).
Next, sodium chromate reacts with dilute acid and hydrogen peroxide. Under mildly acidic conditions the chromate ion forms a blue peroxo complex, commonly written as $$CrO_5$$ (chromium(VI) peroxide):
Ionic form of the reaction:
$$CrO_4^{2-} + 2H^+ + 2H_2O_2 \rightarrow CrO_5 + 3H_2O$$
Adding the spectator sodium and chloride ions gives the required molecular
equation:
$$Na_2CrO_4 + 2HCl + 2H_2O_2 \rightarrow CrO_5 + 2NaCl + 3H_2O$$
Hence, $$B = CrO_5$$.
Therefore
$$A = Na_2CrO_4,\qquad B = CrO_5$$, which corresponds to Option A.
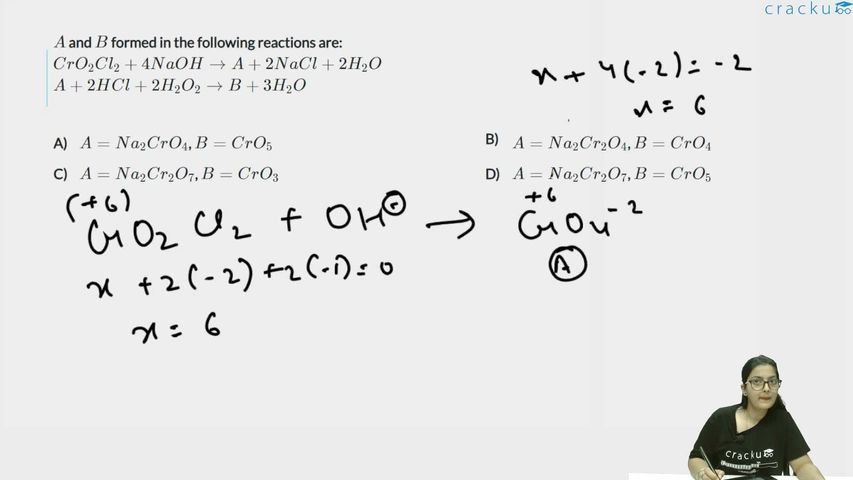
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
